QUESTION IMAGE
Question
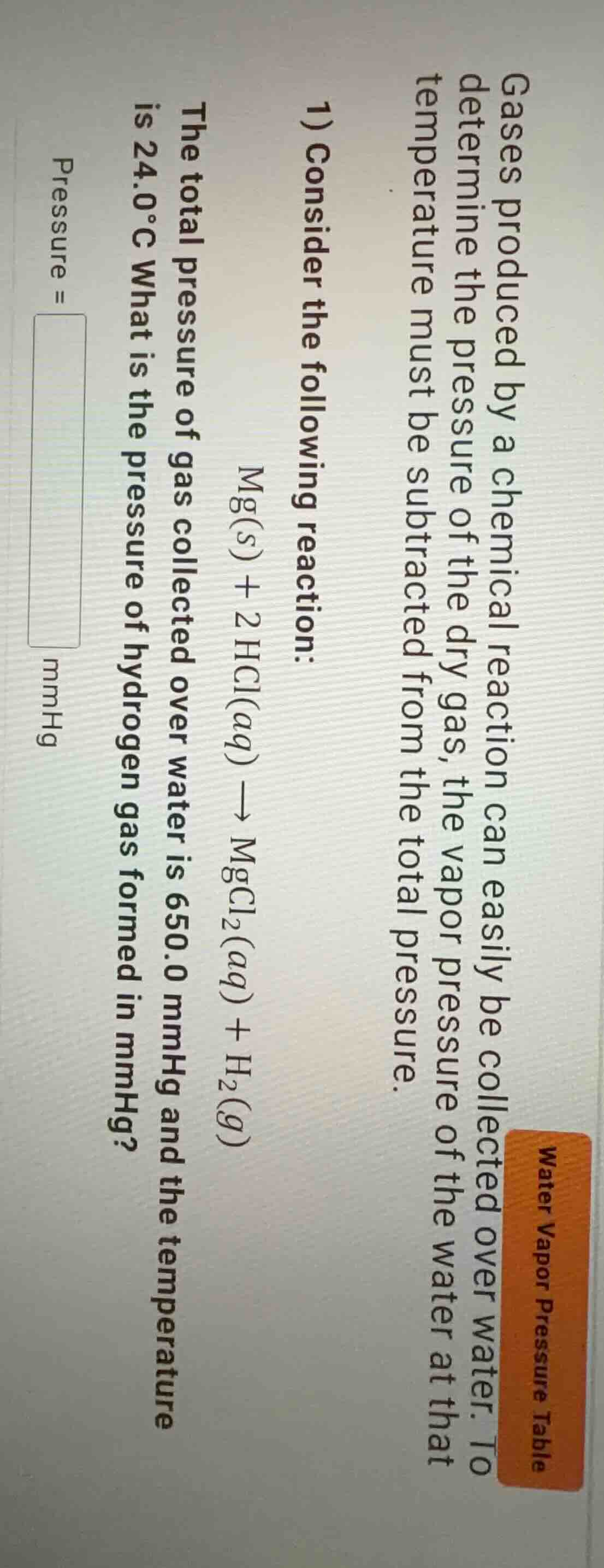
gases produced by a chemical reaction can easily be collected over water. to determine the pressure of the dry gas, the vapor pressure of the water at that temperature must be subtracted from the total pressure.
- consider the following reaction:
\ce{mg(s) + 2hcl(aq) -> mgcl2(aq) + h2(g)}
the total pressure of gas collected over water is 650.0 mmhg and the temperature is 24.0°c what is the pressure of hydrogen gas formed in mmhg?
pressure = \boxed{} mmhg
water vapor pressure table
Step1: Recall water vapor pressure at 24.0°C
From water vapor pressure tables, at \( T = 24.0^\circ\text{C} \), the vapor pressure of water (\( P_{\text{H}_2\text{O}} \)) is 22.4 mmHg (standard value for this temperature).
Step2: Apply Dalton's Law of Partial Pressures
Dalton's Law states that the total pressure (\( P_{\text{total}} \)) is the sum of the partial pressures of the individual gases:
\( P_{\text{total}} = P_{\text{H}_2} + P_{\text{H}_2\text{O}} \)
We need to solve for \( P_{\text{H}_2} \):
\( P_{\text{H}_2} = P_{\text{total}} - P_{\text{H}_2\text{O}} \)
Given \( P_{\text{total}} = 650.0 \, \text{mmHg} \) and \( P_{\text{H}_2\text{O}} = 22.4 \, \text{mmHg} \), substitute the values:
\( P_{\text{H}_2} = 650.0 - 22.4 \)
Step3: Calculate the result
\( 650.0 - 22.4 = 627.6 \, \text{mmHg} \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( 627.6 \) mmHg