QUESTION IMAGE
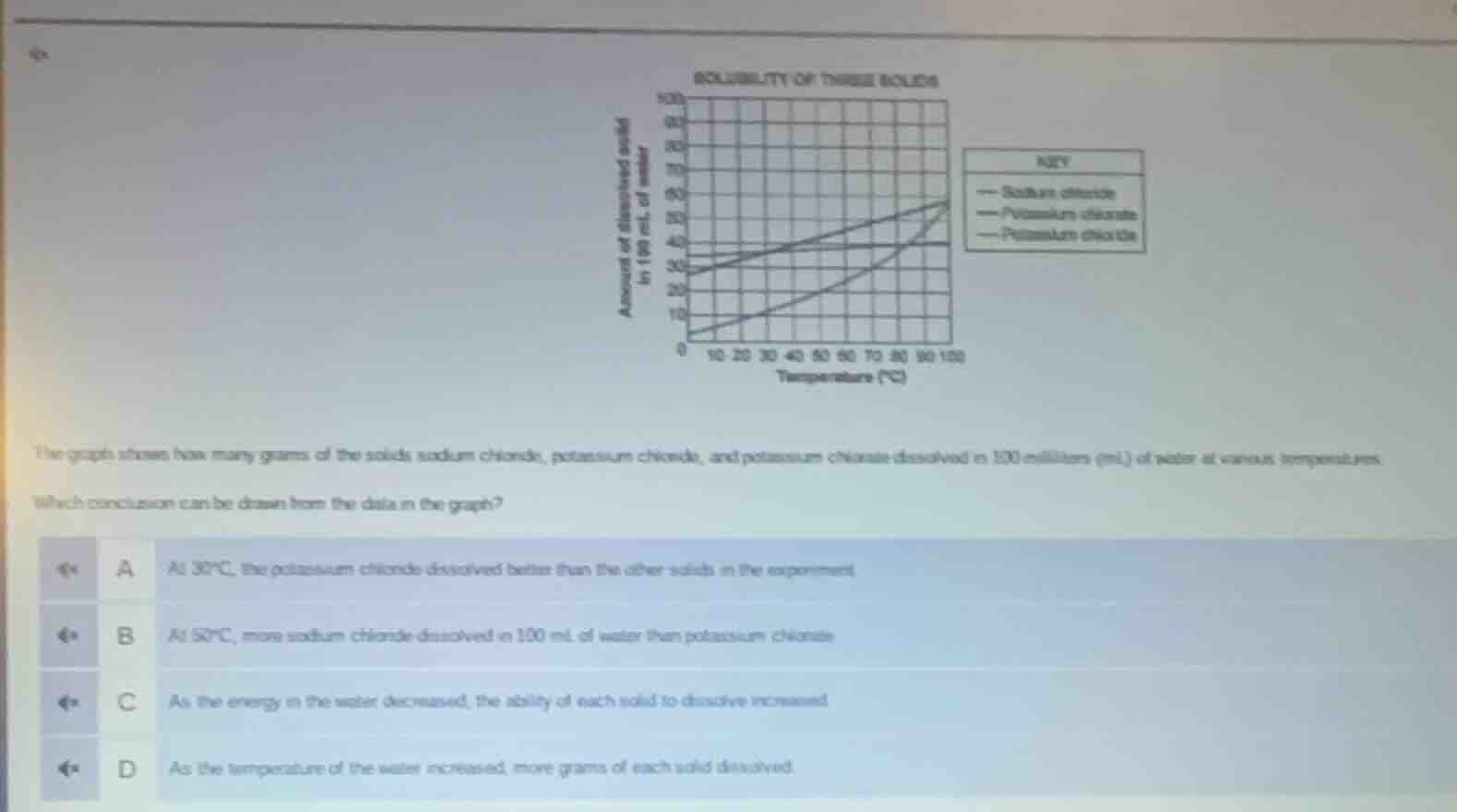
Question
the graph shows how many grams of the solids sodium chloride, potassium chloride, and potassium chlorate dissolved in 100 milliliters (ml) of water at various temperatures.
which conclusion can be drawn from the data in the graph?
a at 30°c, the potassium chloride dissolved better than the other solids in the experiment
b at 50°c, more sodium chloride dissolved in 100 ml of water than potassium chlorate
c as the energy in the water decreased, the ability of each solid to dissolve increased
d as the temperature of the water increased, more grams of each solid dissolved
- Analyze Option A: At 30°C, the solubility of sodium chloride (~36g) is higher than potassium chloride (~32g), so potassium chloride does not dissolve better than all others. This is incorrect.
- Analyze Option B: At 50°C, the solubility of sodium chloride (~37g) is higher than potassium chloride (~40g? No, correction: looking at the graph, at 50°C, sodium chloride is ~37g, potassium chloride is ~40g? No, wait, no—wait the lines: sodium chloride is the top flat line, potassium chloride is the middle rising line, potassium chlorate is the bottom rising line. At 50°C, sodium chloride (~37g) is higher than potassium chloride (~40g? No, no, the y-axis is amount dissolved. Wait, no, at 50°C, sodium chloride is ~37g, potassium chloride is ~40g? No, no, the line for sodium chloride is almost flat, starting at ~30g at 0°C, going to ~38g at 100°C. Potassium chloride starts at ~28g at 0°C, goes to ~55g at 100°C. At 50°C, sodium chloride is ~37g, potassium chloride is ~40g? No, that can't be. Wait, no, the question says Option B: "At 50°C, more sodium chloride dissolved in 100 mL of water than potassium chloride." If at 50°C, sodium chloride is ~37g, potassium chloride is ~40g, then B is wrong. Wait, no, maybe I misread the lines. Wait the key: Sodium chloride is the top line (starts at ~30, goes to ~38), Potassium chloride is the middle line (starts at ~28, goes to ~55), Potassium chlorate is the bottom line (starts at ~5, goes to ~55). At 30°C, sodium chloride is ~36g, potassium chloride is ~32g, so A is wrong. At 50°C, sodium chloride is ~37g, potassium chloride is ~40g? No, that would mean B is wrong. Wait Option D: "As the temperature of the water increased, more grams of each solid dissolved." Looking at the graph, all three lines have a positive slope (increase from left to right), meaning solubility increases with temperature for all three. Option C: "As the energy in the water decreased, the ability of each solid to dissolve increased." Since lower temperature means lower water energy, and solubility decreases with lower temperature, this is wrong. Wait Option B: maybe I misread. Wait at 50°C, sodium chloride is ~37g, potassium chloride is ~40g? No, that would mean B is wrong. Wait no, maybe the lines are: Sodium chloride is the middle line? No, the key says: Sodium chloride (top line, flat), Potassium chlorate (bottom line, steep), Potassium chloride (middle line, moderate slope). At 50°C, sodium chloride is ~37g, potassium chloride is ~40g? No, that would mean B is wrong. Wait Option D says "As the temperature of the water increased, more grams of each solid dissolved." All three lines go up as temperature increases, so this is correct. Wait but let's recheck Option B: "At 50°C, more sodium chloride dissolved in 100 mL of water than potassium chloride." If at 50°C, sodium chloride is ~37g, potassium chloride is ~40g, then B is wrong. Option A: At 30°C, potassium chloride (~32g) is less than sodium chloride (~36g), so A is wrong. Option C: Decreased water energy (lower temp) leads to decreased solubility, so C is wrong. Option D is correct because all three solubility curves show an increase with temperature.
Wait, no, wait the sodium chloride line is almost flat, but it does increase slightly from ~30g at 0°C to ~38g at 100°C, so it does increase with temperature. So all three solids show increased solubility with increasing temperature.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. As the temperature of the water increased, more grams of each solid dissolved.