QUESTION IMAGE
Question
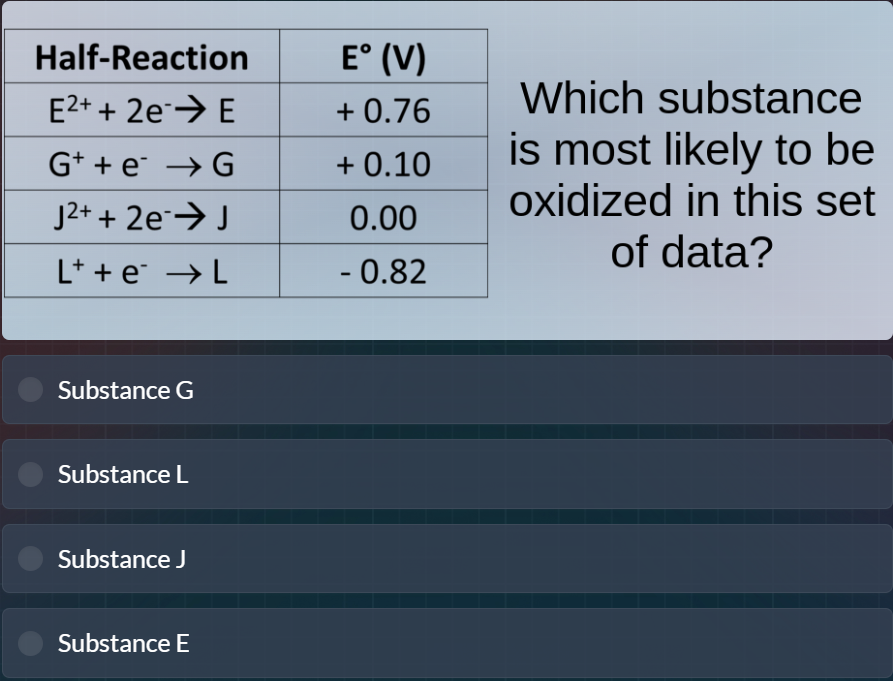
half-reaction | e° (v)
e²⁺ + 2e⁻→ e | + 0.76
g⁺ + e⁻ → g | + 0.10
j²⁺ + 2e⁻→ j | 0.00
l⁺ + e⁻ → l | - 0.82
which substance is most likely to be oxidized in this set of data?
substance g
substance l
substance j
substance e
To determine which substance is most likely to be oxidized, we use the concept of standard reduction potentials (\(E^\circ\)). A substance that is easily oxidized is a strong reducing agent, meaning it has a lower (more negative) standard reduction potential because oxidation is the reverse of reduction.
- For \(E^{2+} + 2e^-
ightarrow E\), \(E^\circ = +0.76\) V (reduction, so \(E\) is a weak reducing agent as it prefers to be reduced).
- For \(G^+ + e^-
ightarrow G\), \(E^\circ = +0.10\) V (reduction, \(G\) is less likely to be oxidized than those with lower \(E^\circ\)).
- For \(J^{2+} + 2e^-
ightarrow J\), \(E^\circ = 0.00\) V (reduction, \(J\) is less likely to be oxidized than those with more negative \(E^\circ\)).
- For \(L^+ + e^-
ightarrow L\), \(E^\circ = -0.82\) V (reduction; the reverse reaction, oxidation of \(L\) to \(L^+\), is more favorable because a very negative \(E^\circ\) for reduction means the forward reduction is unfavorable, so the reverse (oxidation) is favorable).
Among the substances, \(L\) has the most negative \(E^\circ\) for reduction, so it is the most likely to be oxidized (act as a reducing agent by losing electrons).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Substance L