QUESTION IMAGE
Question
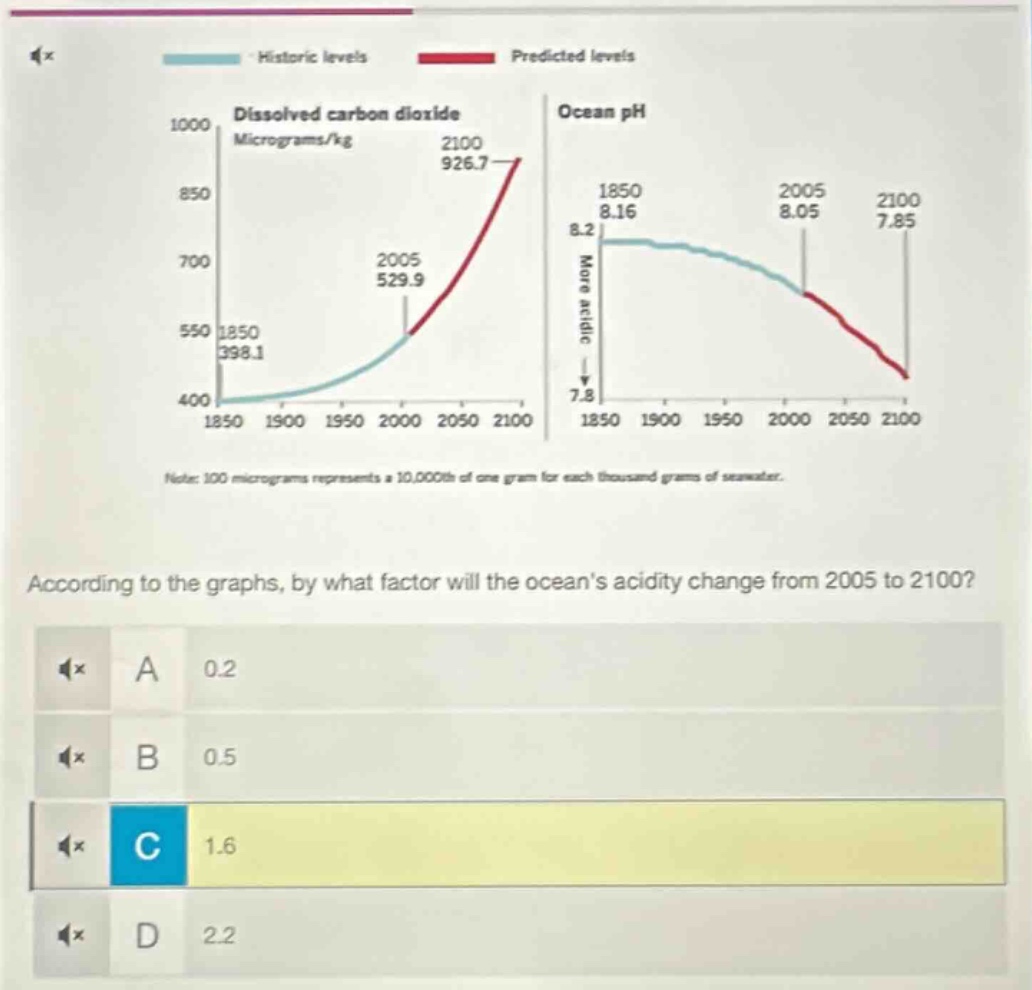
historic levels predicted levels dissolved carbon dioxide ocean ph micrograms/kg 1000 2100 926.7 850 1850 2005 8.16 8.05 2100 7.85 700 2005 529.9 8.2 more acidic 550 1850 398.1 7.8 400 1850 1900 1950 2000 2050 2100 1850 1900 1950 2000 2050 2100 note: 100 micrograms represents a 10,000th of one gram for each thousand grams of seawater. according to the graphs, by what factor will the oceans acidity change from 2005 to 2100? a 0.2 b 0.5 c 1.6 d 2.2
Step1: Recall pH-acidity relationship
Acidity is $[\text{H}^+]$, and $\text{pH} = -\log_{10}[\text{H}^+]$, so $[\text{H}^+] = 10^{-\text{pH}}$.
Step2: Calculate 2005 hydrogen ion concentration
2005 ocean pH = 8.05, so $[\text{H}^+]_{2005} = 10^{-8.05}$
Step3: Calculate 2100 hydrogen ion concentration
2100 ocean pH = 7.85, so $[\text{H}^+]_{2100} = 10^{-7.85}$
Step4: Find the acidity factor
Factor = $\frac{[\text{H}^+]_{2100}}{[\text{H}^+]_{2005}} = \frac{10^{-7.85}}{10^{-8.05}} = 10^{(-7.85 + 8.05)} = 10^{0.2} \approx 1.6$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. 1.6