QUESTION IMAGE
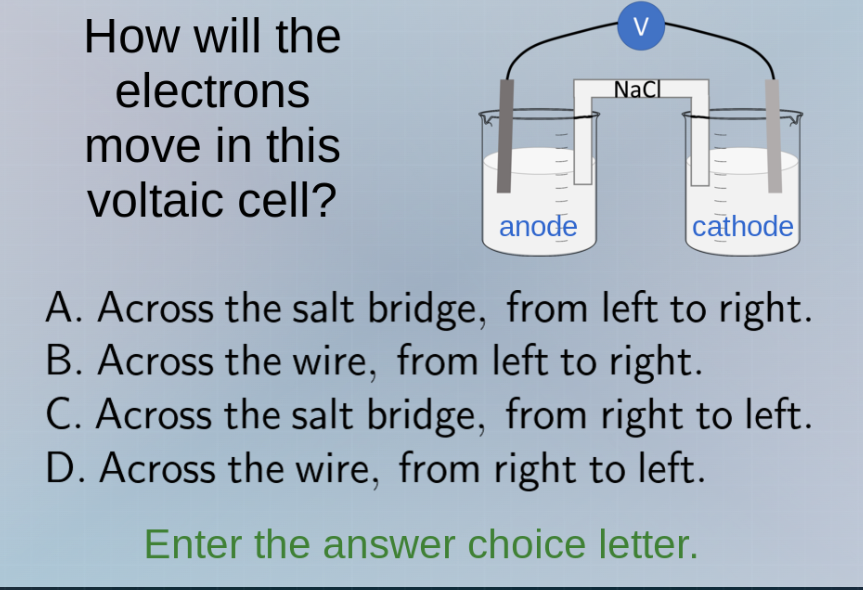
Question
how will the electrons move in this voltaic cell? a. across the salt bridge, from left to right. b. across the wire, from left to right. c. across the salt bridge, from right to left. d. across the wire, from right to left. enter the answer choice letter.
In a voltaic cell, electrons flow from the anode (where oxidation occurs, releasing electrons) to the cathode (where reduction occurs, accepting electrons) through the external wire. The anode is on the left, cathode on the right. So electrons move across the wire from left (anode) to right (cathode)? Wait, no—wait, anode is oxidation (loses electrons), cathode is reduction (gains electrons). So electron flow is from anode to cathode. But in the diagram, anode is left, cathode right. Wait, but electron flow direction: in the wire, electrons move from anode (left) to cathode (right)? Wait no, wait the conventional current is opposite, but electron flow is from negative (anode, since it's releasing electrons, so it's the negative electrode in voltaic cell? Wait no, in voltaic cell, anode is negative, cathode positive. So electrons, being negative, move from anode (left) to cathode (right) through the wire. Wait but the options: B is across wire from left to right, D is right to left. Wait maybe I got it reversed. Wait, no: in a voltaic cell, the anode is where oxidation happens (electrons are produced), so electrons accumulate at the anode, making it negative. The cathode is where reduction happens (electrons are consumed), so it's positive. So electrons flow from the negative anode (left) to positive cathode (right) through the external wire. So the wire is the external circuit. The salt bridge allows ions to move, not electrons. So electrons move through the wire, from anode (left) to cathode (right)? Wait but the options: B is across wire from left to right. Wait but maybe I made a mistake. Wait, no—wait the anode is on the left, cathode on the right. So electrons leave the anode (left) and go to the cathode (right) through the wire. So the direction is from left (anode) to right (cathode) in the wire. So option B? Wait no, wait maybe the other way. Wait, no, let's recall: in a voltaic cell, the anode is the negative electrode, cathode positive. Electrons flow from negative to positive, so from anode (left) to cathode (right) through the wire. So the wire is the path for electrons. So the correct option is B? Wait no, wait the answer might be D. Wait, maybe I confused the direction. Wait, let's think again. The anode is where oxidation occurs: \( \text{Oxidation: } \text{X}
ightarrow \text{X}^{n+} + ne^- \), so electrons are released at the anode. The cathode is where reduction occurs: \( \text{Reduction: } \text{Y}^{m+} + me^-
ightarrow \text{Y} \), so electrons are consumed at the cathode. So the electrons produced at the anode (left) need to go to the cathode (right) to be consumed. So the flow of electrons is from anode (left) to cathode (right) through the wire. So the wire is the external circuit. So option B: across the wire, from left to right. Wait but maybe I got it wrong. Wait, no—wait the anode is on the left, cathode on the right. So electrons move from left (anode) to right (cathode) in the wire. So the correct option is B? Wait but the options: B is "Across the wire, from left to right." So the answer is B? Wait no, wait maybe the other way. Wait, maybe the anode is the right? No, the diagram shows anode on left, cathode on right. So I think the correct answer is B. Wait but let's check again. The salt bridge is for ions, not electrons. So electrons move through the wire. So from anode (left) to cathode (right) in the wire. So option B. So the answer is B.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B