QUESTION IMAGE
Question
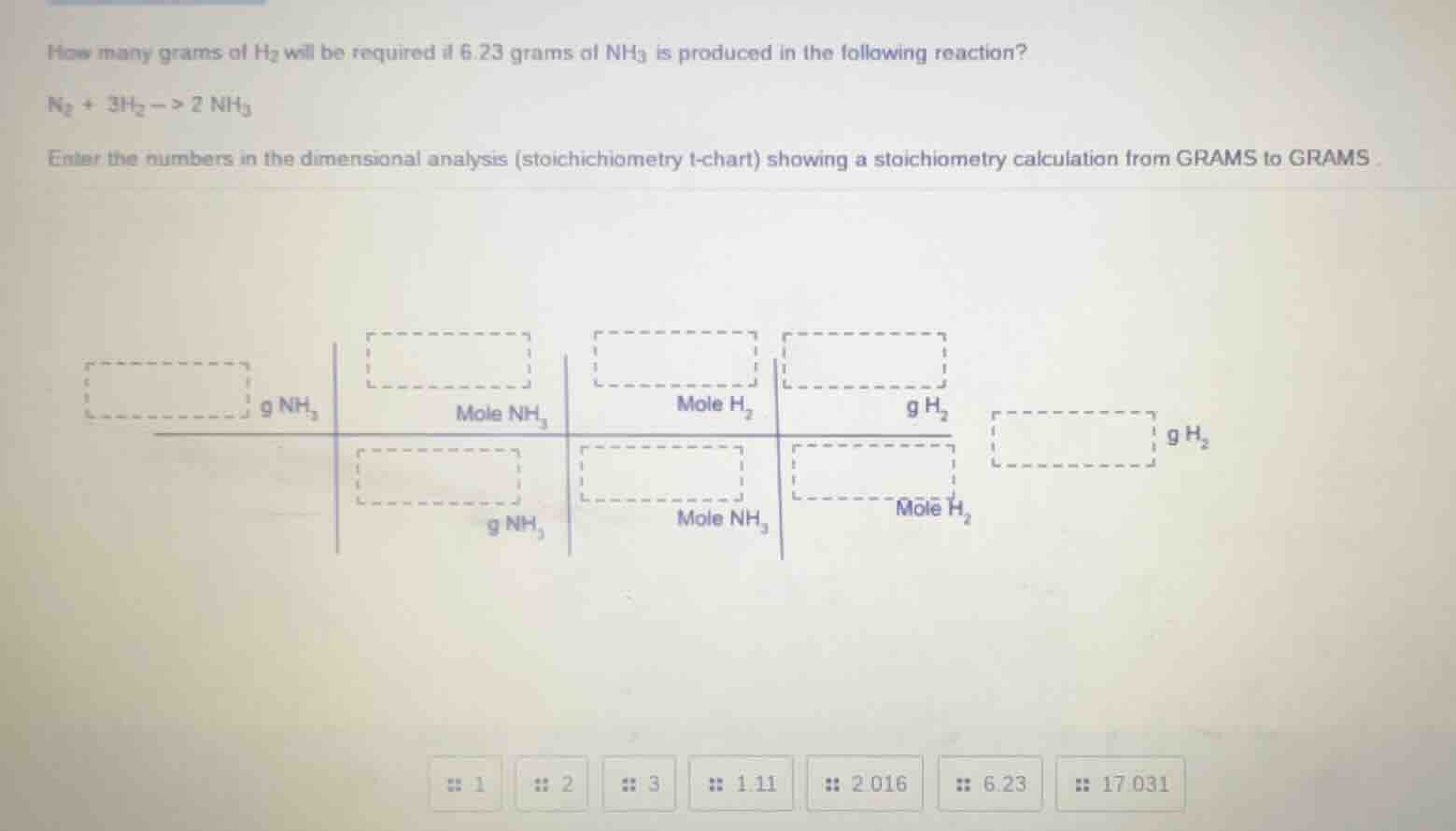
how many grams of h₂ will be required if 6.23 grams of nh₃ is produced in the following reaction?
n₂ + 3h₂ → 2nh₃
enter the numbers in the dimensional analysis (stoichichiometry t-chart) showing a stoichiometry calculation from grams to grams
chart structure: g nh₃, mole nh₃, mole h₂, g h₂, g h₂ in the final box; denominators: g nh₃, mole nh₃, mole h₂
options: 1, 2, 3, 1.11, 2.016, 6.23, 17.031
Step1: Start with given mass of NH₃
$6.23\ \text{g NH}_3$
Step2: Convert NH₃ g to moles
Use molar mass of $\text{NH}_3$: $M_{\text{NH}_3}=17.031\ \text{g/mol}$
$\frac{1\ \text{mol NH}_3}{17.031\ \text{g NH}_3}$
Step3: Mole ratio of H₂ to NH₃
From reaction: $3\ \text{mol H}_2 : 2\ \text{mol NH}_3$
$\frac{3\ \text{mol H}_2}{2\ \text{mol NH}_3}$
Step4: Convert H₂ moles to grams
Molar mass of $\text{H}_2$: $M_{\text{H}_2}=2.016\ \text{g/mol}$
$\frac{2.016\ \text{g H}_2}{1\ \text{mol H}_2}$
Step5: Calculate final mass
$6.23\ \text{g NH}_3 \times \frac{1\ \text{mol NH}_3}{17.031\ \text{g NH}_3} \times \frac{3\ \text{mol H}_2}{2\ \text{mol NH}_3} \times \frac{2.016\ \text{g H}_2}{1\ \text{mol H}_2}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Filled t-chart values (left to right, top then bottom):
Top row: 1, 3, 2.016
Bottom row: 17.031, 2, 1
Final calculated mass of $\text{H}_2$: $1.11\ \text{g}$
(The completed dimensional analysis is:
$6.23\ \text{g NH}_3 \times \frac{1\ \text{mol NH}_3}{17.031\ \text{g NH}_3} \times \frac{3\ \text{mol H}_2}{2\ \text{mol NH}_3} \times \frac{2.016\ \text{g H}_2}{1\ \text{mol H}_2} = 1.11\ \text{g H}_2$)