QUESTION IMAGE
Question
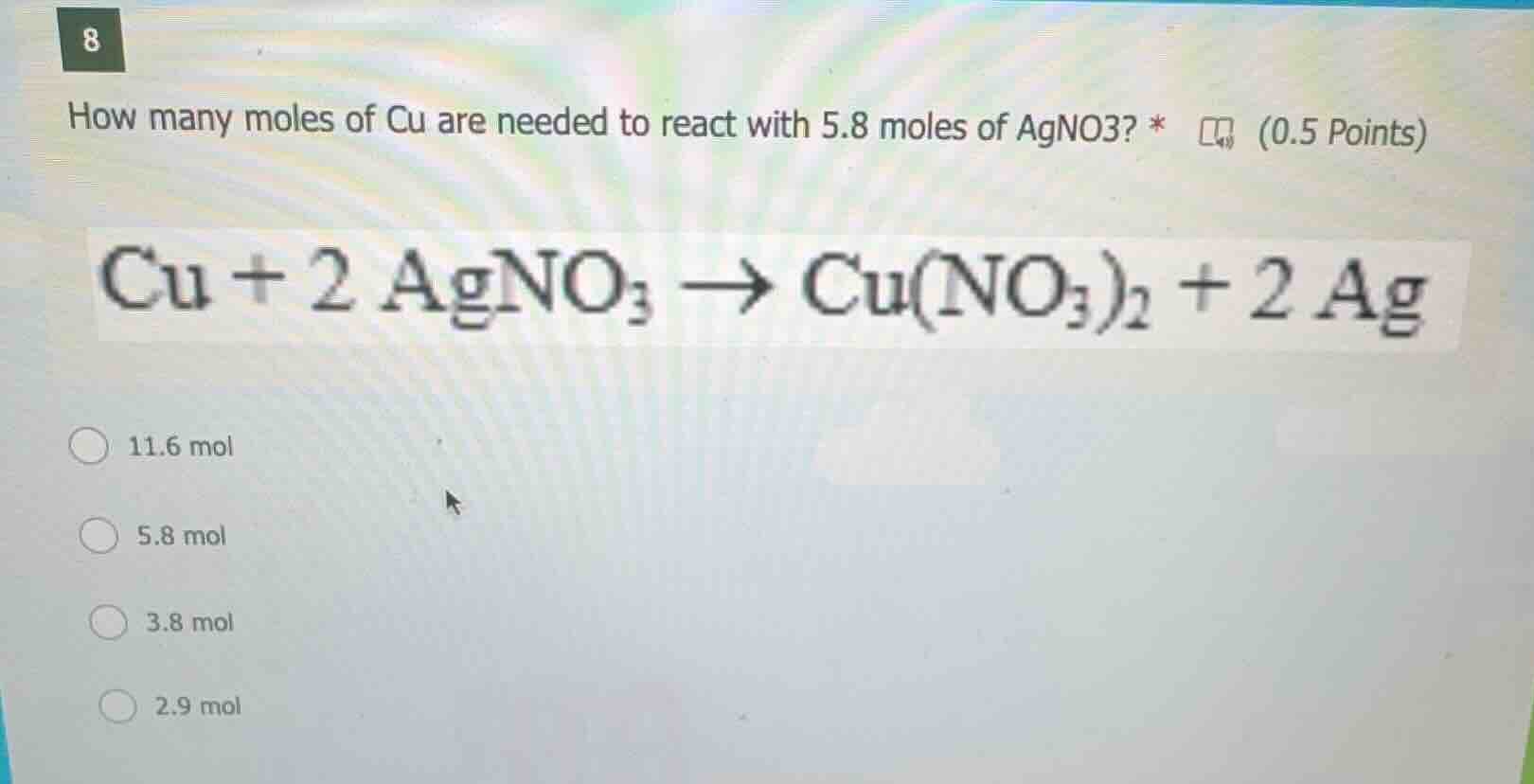
8
how many moles of cu are needed to react with 5.8 moles of agno3? *
cu + 2 agno₃ → cu(no₃)₂ + 2 ag
11.6 mol
5.8 mol
3.8 mol
2.9 mol
Step1: Identify mole ratio
From the reaction \( \text{Cu} + 2\text{AgNO}_3
ightarrow \text{Cu(NO}_3\text{)}_2 + 2\text{Ag} \), the mole ratio of \( \text{Cu} \) to \( \text{AgNO}_3 \) is \( 1:2 \).
Step2: Calculate moles of Cu
Let \( n(\text{Cu}) \) be moles of Cu. Using the ratio: \( \frac{n(\text{Cu})}{n(\text{AgNO}_3)} = \frac{1}{2} \). Given \( n(\text{AgNO}_3) = 5.8 \) mol, so \( n(\text{Cu}) = \frac{1}{2} \times 5.8 = 2.9 \) mol.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
2.9 mol (corresponding to the option "2.9 mol")