QUESTION IMAGE
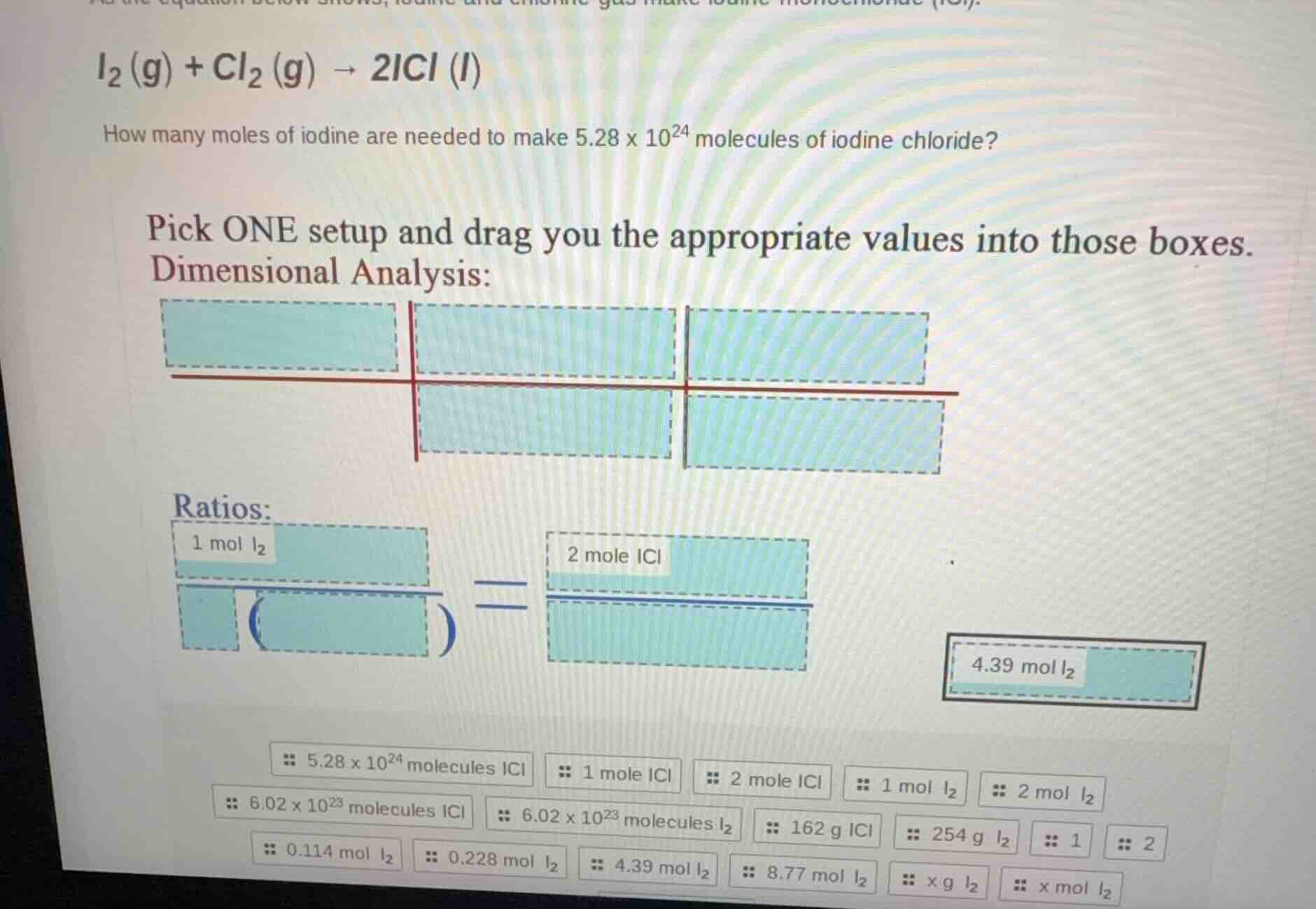
Question
how many moles of iodine are needed to make $5.28 \times 10^{24}$ molecules of iodine chloride?
pick one setup and drag you the appropriate values into those boxes.
dimensional analysis:
boxes
ratios:
$$\frac{1\text{ mol }i_2}{box (box)} = \frac{2\text{ mole }icl}{box}$$
4.39 mol $i_2$
5.28 × 10²⁴ molecules icl 1 mole icl 2 mole icl 1 mol $i_2$ 2 mol $i_2$
6.02 × 10²³ molecules icl 6.02 × 10²³ molecules $i_2$ 162 g icl 254 g $i_2$ 1 2
0.114 mol $i_2$ 0.228 mol $i_2$ 4.39 mol $i_2$ 8.77 mol $i_2$ x g $i_2$ x mol $i_2$
Step1: Convert ICl molecules to moles
Use Avogadro's number to convert the number of ICl molecules to moles.
$\text{Moles of ICl} = \frac{5.28 \times 10^{24} \text{ molecules ICl}}{6.02 \times 10^{23} \text{ molecules ICl/mol ICl}}$
Step2: Use mole ratio to find $\text{I}_2$ moles
From the balanced equation, 1 mol $\text{I}_2$ produces 2 mol ICl.
$\text{Moles of } \text{I}_2 = \frac{5.28 \times 10^{24}}{6.02 \times 10^{23}} \times \frac{1 \text{ mol } \text{I}_2}{2 \text{ mol ICl}}$
Step3: Calculate the final value
Compute the numerical result.
$\text{Moles of } \text{I}_2 = \frac{5.28 \times 10^{24}}{6.02 \times 10^{23} \times 2} \approx 4.39$
Dimensional Analysis Setup:
Top row (left to right): $5.28 \times 10^{24} \text{ molecules ICl}$, $1 \text{ mol ICl}$, $1 \text{ mol } \text{I}_2$
Bottom row (left to right): $6.02 \times 10^{23} \text{ molecules ICl}$, $2 \text{ mol ICl}$
Ratios Setup:
$\frac{1 \text{ mol } \text{I}_2}{2 \text{ mol ICl}} = \frac{x \text{ mol } \text{I}_2}{(\frac{5.28 \times 10^{24}}{6.02 \times 10^{23}}) \text{ mol ICl}}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$4.39 \text{ mol } \text{I}_2$