QUESTION IMAGE
Question
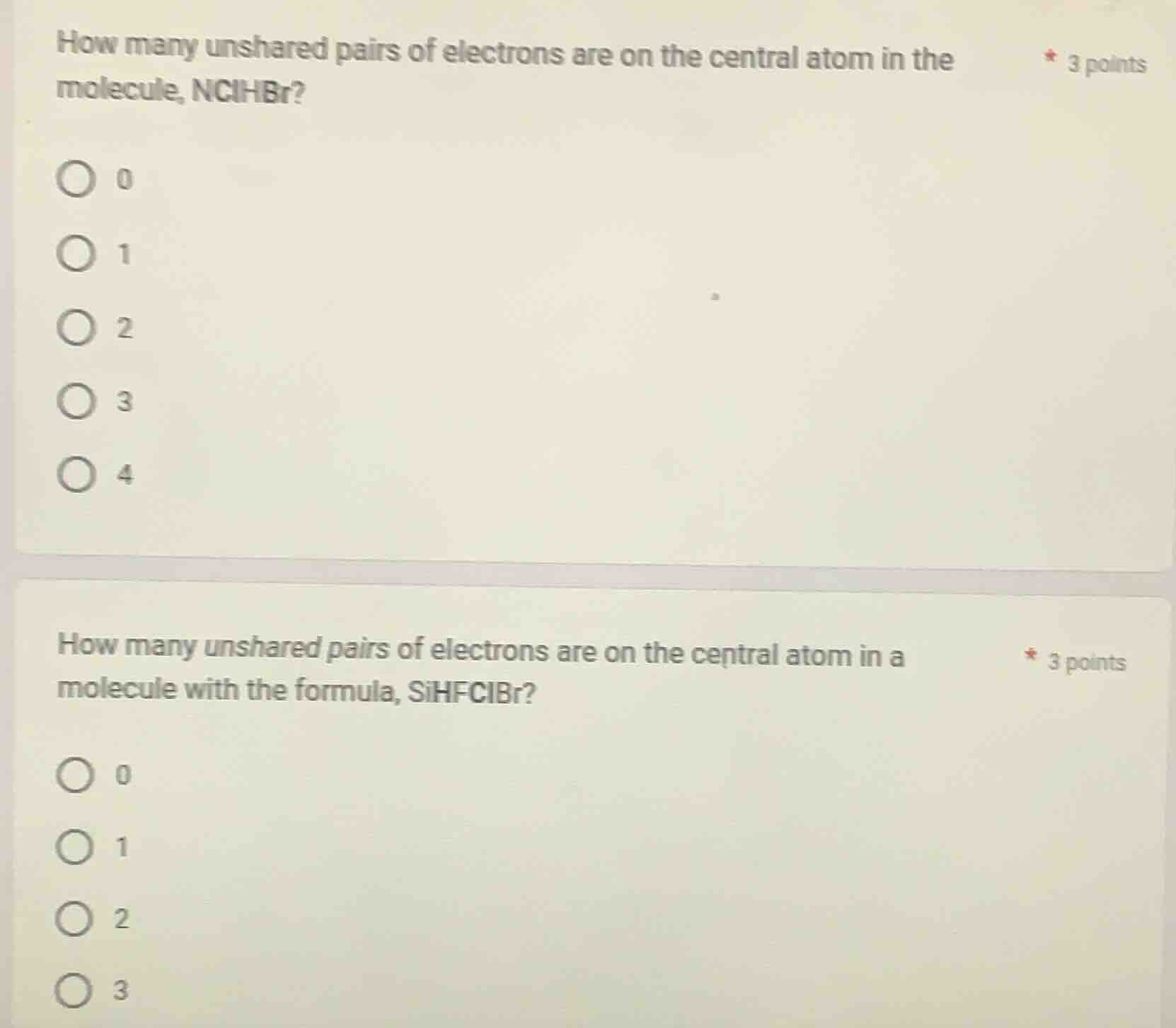
how many unshared pairs of electrons are on the central atom in the molecule, nclhbr? 3 points 0 1 2 3 4 how many unshared pairs of electrons are on the central atom in a molecule with the formula, sihfclbr? 3 points 0 1 2 3
Step1: Analyze NClHBr central atom
Nitrogen (N) has 5 valence electrons. It forms 3 single bonds (to Cl, H, Br), using 6 electrons. Calculate remaining electrons: $5 - 3 = 2$ electrons = 1 unshared pair.
Step2: Analyze SiHFClBr central atom
Silicon (Si) has 4 valence electrons. It forms 4 single bonds (to H, F, Cl, Br), using all 4 valence electrons. Remaining electrons: $4 - 4 = 0$ electrons = 0 unshared pairs.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- For NClHBr: 1
- For SiHFClBr: 0