QUESTION IMAGE
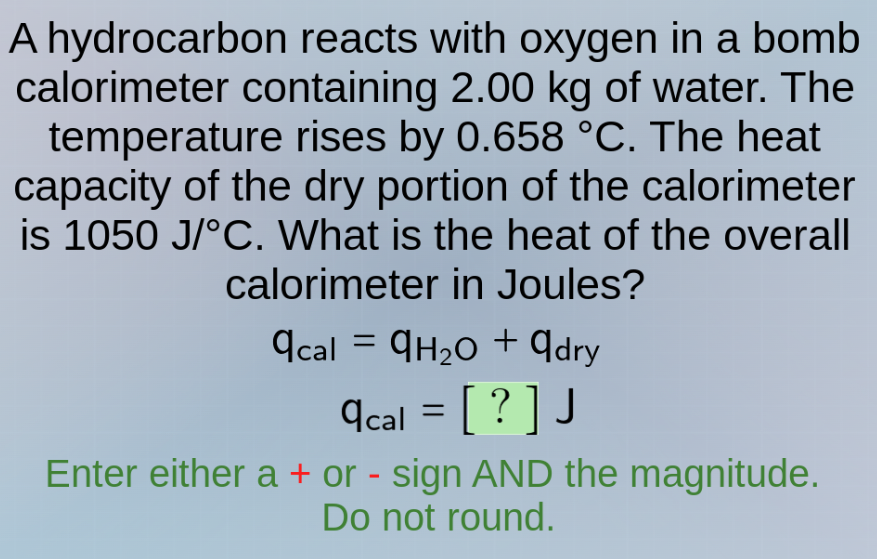
Question
a hydrocarbon reacts with oxygen in a bomb calorimeter containing 2.00 kg of water. the temperature rises by 0.658 °c. the heat capacity of the dry portion of the calorimeter is 1050 j/°c. what is the heat of the overall calorimeter in joules?
q_cal = q_h₂o + q_dry
q_cal = ? j
enter either a + or - sign and the magnitude.
do not round.
Step1: Calculate \( q_{H_2O} \)
The formula for heat absorbed by water is \( q = mc\Delta T \). The mass of water \( m = 2.00 \, \text{kg} = 2000 \, \text{g} \), specific heat of water \( c = 4.184 \, \text{J/g}^\circ\text{C} \), and \( \Delta T = 0.658^\circ\text{C} \).
\( q_{H_2O} = 2000 \, \text{g} \times 4.184 \, \text{J/g}^\circ\text{C} \times 0.658^\circ\text{C} \)
Step2: Calculate \( q_{dry} \)
The formula for heat absorbed by the dry portion is \( q = C\Delta T \), where \( C = 1050 \, \text{J/}^\circ\text{C} \) and \( \Delta T = 0.658^\circ\text{C} \).
\( q_{dry} = 1050 \, \text{J/}^\circ\text{C} \times 0.658^\circ\text{C} \)
Step3: Calculate \( q_{cal} \)
Using \( q_{cal} = q_{H_2O} + q_{dry} \)
First, compute \( q_{H_2O} \):
\( 2000 \times 4.184 \times 0.658 = 2000 \times 2.753072 = 5506.144 \)
Then, compute \( q_{dry} \):
\( 1050 \times 0.658 = 690.9 \)
Now, sum them: \( q_{cal} = 5506.144 + 690.9 = 6197.044 \)
Since the temperature rises, the calorimeter absorbs heat, so \( q_{cal} \) is positive.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
+6197.044