QUESTION IMAGE
Question
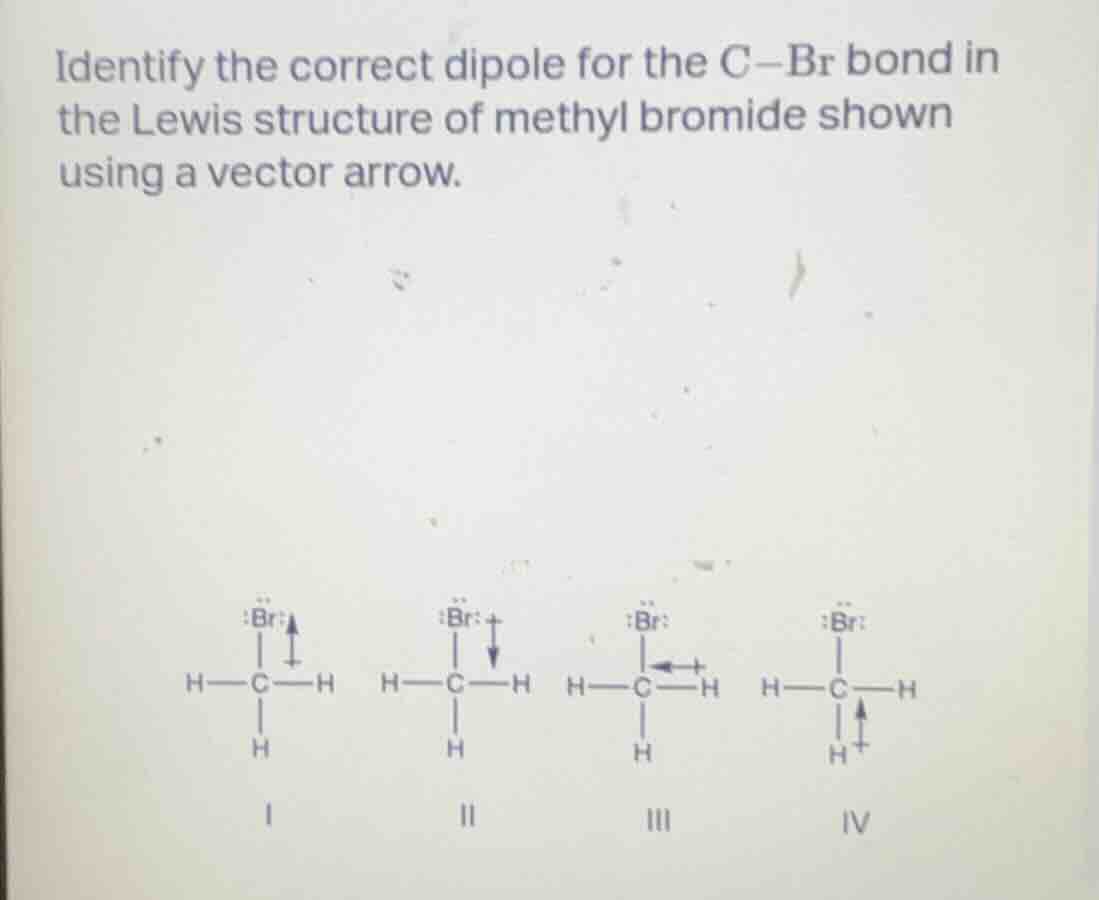
identify the correct dipole for the c$-$br bond in the lewis structure of methyl bromide shown using a vector arrow.
i
ii
iii
iv
Brief Explanations
Dipole arrows point from the less electronegative atom to the more electronegative atom. Bromine (Br) is more electronegative than carbon (C), so the arrow should start at C and point toward Br, with the positive end (+) on C and the arrowhead on Br.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
II. $\ce{H - \underset{\underset{H}{|}}{C} - H}$ with arrow pointing from C to Br (positive end on C, arrowhead on Br)