QUESTION IMAGE
Question
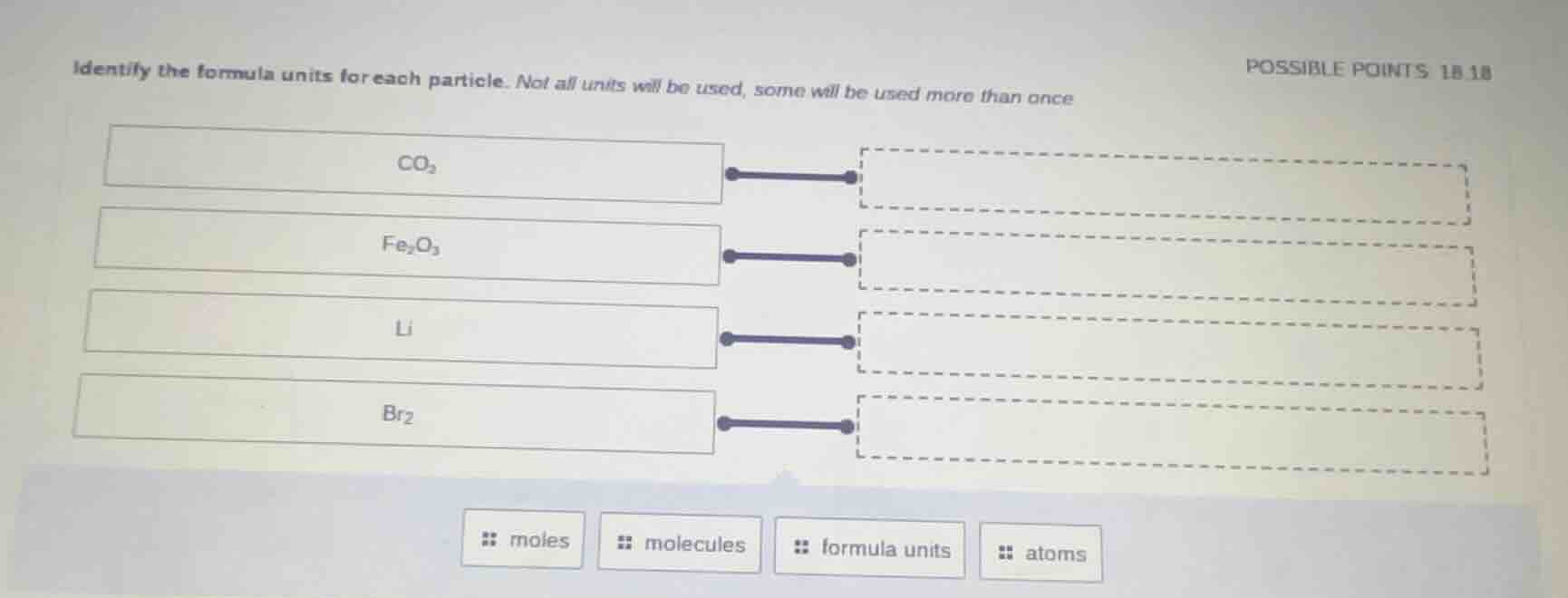
identify the formula units for each particle. not all units will be used, some will be used more than once
co₂
fe₂o₃
li
br₂
moles molecules formula units atoms
Step1: Analyze \( \text{CO}_2 \)
\( \text{CO}_2 \) is a covalent compound (molecular compound). The particle units for molecular compounds can be molecules (when referring to individual discrete units) or moles (amount), but for the formula unit type, for molecular compounds, the formula unit is also the molecular formula, and we can refer to their particles as molecules (or moles, but in terms of the particle type here, \( \text{CO}_2 \) particles are molecules (or can be in moles, but let's match the options). Wait, actually, for \( \text{CO}_2 \):
- Moles: can be used (amount), but the particle type (the formula unit's particle) for molecular compounds: \( \text{CO}_2 \) particles are molecules. But also, moles can be applied to any substance. Wait, maybe better to recall:
- Atoms: for single atoms (like Li).
- Molecules: for covalent/molecular compounds (like \( \text{CO}_2 \), \( \text{Br}_2 \)).
- Formula units: for ionic compounds (like \( \text{Fe}_2\text{O}_3 \)).
- Moles: is an amount unit, can be used for any substance (but the question is about the formula units for each particle, so maybe the particle type). Wait, the question says "Identify the formula units for each particle". Wait, maybe rephrasing: what is the particle type (in terms of the options: moles, molecules, formula units, atoms) for each substance.
So:
- \( \text{CO}_2 \): molecular compound, so its particles are molecules (or formula unit is the molecular formula, but the particle type here would be molecules (or moles, but moles is amount). Wait, the options are moles, molecules, formula units, atoms.
Let's clarify each substance:
- \( \text{CO}_2 \): covalent (molecular) compound. The basic particle is a molecule. Also, we can have moles of \( \text{CO}_2 \), but the particle type (the formula unit's particle) – for molecular compounds, the formula unit is the molecule. So \( \text{CO}_2 \) particles can be molecules (or moles, but moles is amount). Wait, maybe the question is about what type of particle (atom, molecule, formula unit) or the unit (moles is amount, but the other options are particle types). Wait, maybe the options are the types of particles or the units to describe the amount? Wait, the options are: moles (amount), molecules (particle), formula units (particle for ionic), atoms (particle).
So:
- \( \text{Li} \): is an element, a metal, exists as atoms (or in metallic lattice, but the particle type is atoms). So \( \text{Li} \) particles are atoms.
- \( \text{Fe}_2\text{O}_3 \): ionic compound (iron(III) oxide), so its formula unit is \( \text{Fe}_2\text{O}_3 \), and the particle type is formula units (since ionic compounds are made of ions, but the formula unit is the simplest ratio, and we refer to their particles as formula units).
- \( \text{Br}_2 \): diatomic molecular element, so its particles are molecules (or atoms? No, \( \text{Br}_2 \) is a molecule made of two Br atoms). So \( \text{Br}_2 \) particles are molecules.
- \( \text{CO}_2 \): molecular compound, particles are molecules.
- \( \text{Fe}_2\text{O}_3 \): ionic, particles are formula units.
- \( \text{Li} \): particle is atom.
- \( \text{Br}_2 \): particle is molecule.
Also, moles is an amount unit, so any substance can be in moles, but the question is about the formula units (the particle type) for each particle. Wait, maybe the question is matching each substance to the appropriate particle type (atoms, molecules, formula units) or moles (but moles is amount). Let's proceed:
- \( \text{CO}_2 \): molecules (or moles, but moles is amount. Wait, the…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- \( \text{CO}_2 \): molecules (or moles, but based on particle type, molecules)
- \( \text{Fe}_2\text{O}_3 \): formula units
- \( \text{Li} \): atoms
- \( \text{Br}_2 \): molecules
(If we consider moles as an amount unit applicable to all, but the typical matching for this type of problem is as above, with \( \text{Li} \) → atoms, \( \text{Fe}_2\text{O}_3 \) → formula units, \( \text{CO}_2 \) and \( \text{Br}_2 \) → molecules.)
To present the final matches (assuming the question is to connect each substance to the correct particle type/unit):
- \( \text{CO}_2 \) → molecules
- \( \text{Fe}_2\text{O}_3 \) → formula units
- \( \text{Li} \) → atoms
- \( \text{Br}_2 \) → molecules
(Note: "moles" is an amount unit that can apply to all, but the other options are particle types, and this is the standard matching for such problems.)