QUESTION IMAGE
Question
intermolecular forces
quiz complete
time remaining
57:23
1 2 3 4 5 6 7 8 9 10
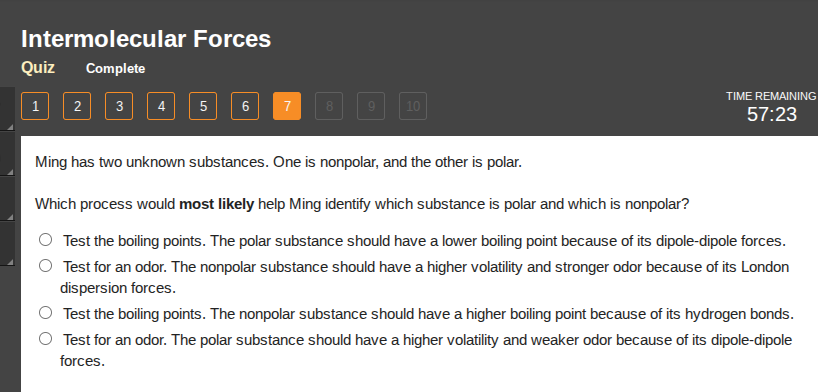
ming has two unknown substances. one is nonpolar, and the other is polar.
which process would most likely help ming identify which substance is polar and which is nonpolar?
test the boiling points. the polar substance should have a lower boiling point because of its dipole-dipole forces.
test for an odor. the nonpolar substance should have a higher volatility and stronger odor because of its london dispersion forces.
test the boiling points. the nonpolar substance should have a higher boiling point because of its hydrogen bonds.
test for an odor. the polar substance should have a higher volatility and weaker odor because of its dipole-dipole forces.
: polar substances have higher boiling points due to stronger forces, nonpolar substances do not form hydrogen bonds, and polar substances have lower volatility.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
: polar substances have higher boiling points due to stronger forces, nonpolar substances do not form hydrogen bonds, and polar substances have lower volatility.