QUESTION IMAGE
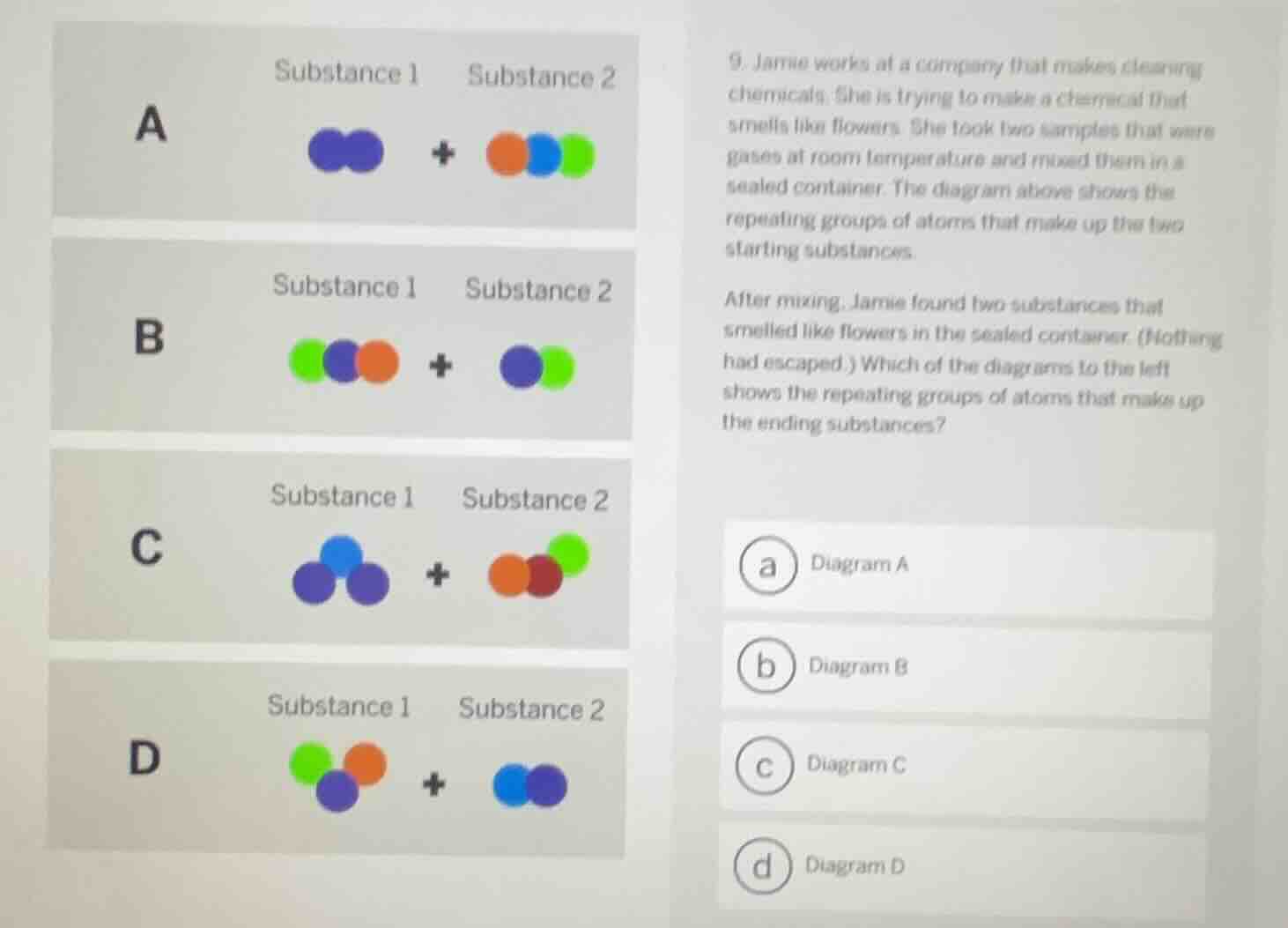
Question
- jamie works at a company that makes cleaning chemicals. she is trying to make a chemical that smells like flowers. she took two samples that were gases at room temperature and mixed them in a sealed container. the diagram above shows the repeating groups of atoms that make up the two starting substances.
after mixing, jamie found two substances that smelled like flowers in the sealed container (nothing had escaped.) which of the diagrams to the left shows the repeating groups of atoms that make up the ending substances?
(a) diagram a
(b) diagram b
(c) diagram c
(d) diagram d
Chemical reactions conserve atoms—no atoms are created or destroyed. The correct diagram should show the same set of atoms from the starting substances rearranged into new repeating groups (molecules) for the two flower-scented products, with no atoms missing or added.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
(Note: The answer cannot be determined without the specific diagram content of the starting substances mentioned in the problem. However, based on the law of conservation of mass, the ending substances must contain the same types and numbers of atoms as the starting substances, just rearranged into new molecules.)