QUESTION IMAGE
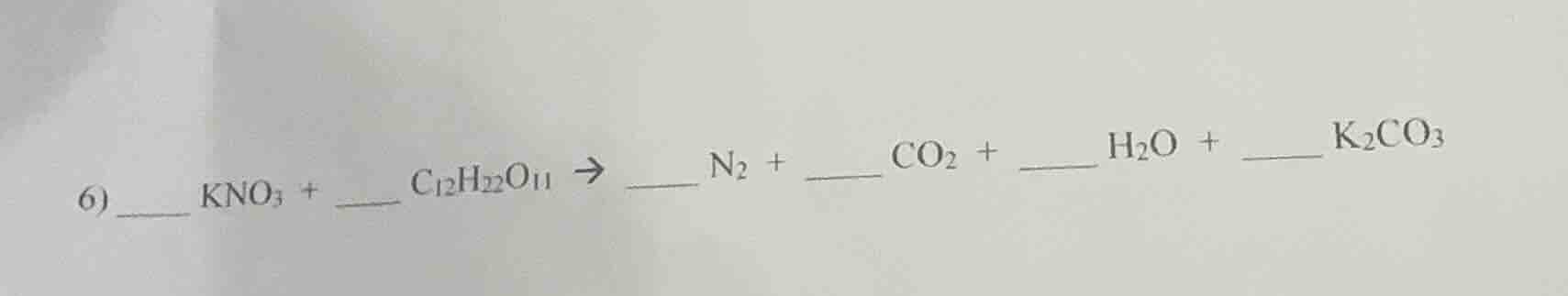
Question
- __ kno₃ + _ c₁₂h₂₂o₁₁ → __ n₂ + co₂ + h₂o + __ k₂co₃
Step1: Identify elements
We have K, N, O, C, H in reactants and products. Let's denote coefficients as \( a \) for \( \text{KNO}_3 \), \( b \) for \( \text{C}_{12}\text{H}_{22}\text{O}_{11} \), \( c \) for \( \text{N}_2 \), \( d \) for \( \text{CO}_2 \), \( e \) for \( \text{H}_2\text{O} \), \( f \) for \( \text{K}_2\text{CO}_3 \).
Step2: Balance K
From K: \( a = 2f \) (since \( \text{KNO}_3 \) has 1 K, \( \text{K}_2\text{CO}_3 \) has 2 K)
Step3: Balance N
From N: \( a = 2c \) ( \( \text{KNO}_3 \) has 1 N, \( \text{N}_2 \) has 2 N)
Step4: Balance C
From C: \( 12b = d + f \) ( \( \text{C}_{12}\text{H}_{22}\text{O}_{11} \) has 12 C, \( \text{CO}_2 \) has 1 C, \( \text{K}_2\text{CO}_3 \) has 1 C)
Step5: Balance H
From H: \( 22b = 2e \) ⇒ \( e = 11b \)
Step6: Balance O
From O: \( 3a + 11b = 2d + e + 3f \)
Let's assume \( b = 1 \) (simplest for sugar). Then:
- From K: \( a = 2f \)
- From N: \( a = 2c \)
- From C: \( 12 = d + f \)
- From H: \( e = 11 \)
- From O: \( 3a + 11 = 2d + 11 + 3f \) ⇒ \( 3a = 2d + 3f \)
Substitute \( d = 12 - f \) and \( a = 2f \) into O balance:
\( 3(2f) = 2(12 - f) + 3f \)
\( 6f = 24 - 2f + 3f \)
\( 6f = 24 + f \)
\( 5f = 24 \)? Wait, no, maybe \( b = 1 \) is not good. Wait, let's try redox approach.
\( \text{KNO}_3 \) is oxidizing agent (N from +5 to 0), \( \text{C}_{12}\text{H}_{22}\text{O}_{11} \) is reducing agent (C from 0 to +4, H from +1 to +1, O from -2 to -2? Wait, C in sugar: formula \( \text{C}_{12}\text{H}_{22}\text{O}_{11} \), oxidation state of C: let x be C's oxidation state. \( 12x + 22(+1) + 11(-2) = 0 \) ⇒ \( 12x + 22 - 22 = 0 \) ⇒ \( x = 0 \). In \( \text{CO}_2 \), C is +4, in \( \text{K}_2\text{CO}_3 \), C is +4. So each C atom loses 4 electrons. There are 12 C atoms, so total electrons lost: \( 12 \times 4 = 48 \).
In \( \text{KNO}_3 \), N is +5, in \( \text{N}_2 \) is 0, so each N gains 5 electrons. There are 2 N atoms per \( \text{N}_2 \), so per \( \text{KNO}_3 \), 1 N gains 5, so per \( \text{N}_2 \), 2 N gain 10 electrons.
Let number of \( \text{N}_2 \) be \( c \), so electrons gained: \( 10c \). Electrons lost: 48 (from 1 \( \text{C}_{12}\text{H}_{22}\text{O}_{11} \)). So \( 10c = 48 \)? No, wait, \( b \) is number of sugar molecules. So electrons lost per \( b \) sugar: \( 12b \times 4 = 48b \). Electrons gained per \( a \) \( \text{KNO}_3 \): each \( \text{KNO}_3 \) has 1 N, so electrons gained per \( \text{KNO}_3 \): 5, so total electrons gained: \( 5a \).
Thus, \( 5a = 48b \). Let's find smallest integers: \( a = 48 \), \( b = 5 \)? Wait, no, maybe I made a mistake. Wait, let's try balancing by trial.
Let's start with \( \text{C}_{12}\text{H}_{22}\text{O}_{11} \) (sucrose) as 1. Then C: 12, so \( d + f = 12 \). H: 22, so \( e = 11 \). K: from \( \text{KNO}_3 \), \( a = 2f \). N: \( a = 2c \). O: \( 3a + 11 = 2d + 11 + 3f \) ⇒ \( 3a = 2d + 3f \). But \( d = 12 - f \), so \( 3a = 2(12 - f) + 3f = 24 + f \). Also, \( a = 2f \), so \( 3(2f) = 24 + f \) ⇒ \( 6f = 24 + f \) ⇒ \( 5f = 24 \) ⇒ \( f = 24/5 \), not integer. So \( b \) must be 5? Wait, no, let's try \( b = 1 \), but adjust. Wait, maybe the reaction is:
\( \text{KNO}_3 + \text{C}_{12}\text{H}_{22}\text{O}_{11}
ightarrow \text{N}_2 + \text{CO}_2 + \text{H}_2\text{O} + \text{K}_2\text{CO}_3 \)
Let's use oxidation states properly.
Oxidation of C: in \( \text{C}_{12}\text{H}_{22}\text{O}_{11} \), C is 0 (as calculated), in \( \text{CO}_2 \) and \( \text{K}_2\text{CO}_3 \), C is +4. So each C loses 4 e⁻, 12 C lose 48 e⁻ per sucrose.
Reduction of N: in \( \text{KNO}_3 \), N is +5, in \( \text{N…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
48 \( \text{KNO}_3 \) + 5 \( \text{C}_{12}\text{H}_{22}\text{O}_{11} \) → 24 \( \text{N}_2 \) + 36 \( \text{CO}_2 \) + 55 \( \text{H}_2\text{O} \) + 24 \( \text{K}_2\text{CO}_3 \)
So the coefficients are 48, 5, 24, 36, 55, 24.