QUESTION IMAGE
Question
lesson 7 - new information and predictions on models
name _________________________
date ____________
exit ticket
passage iv
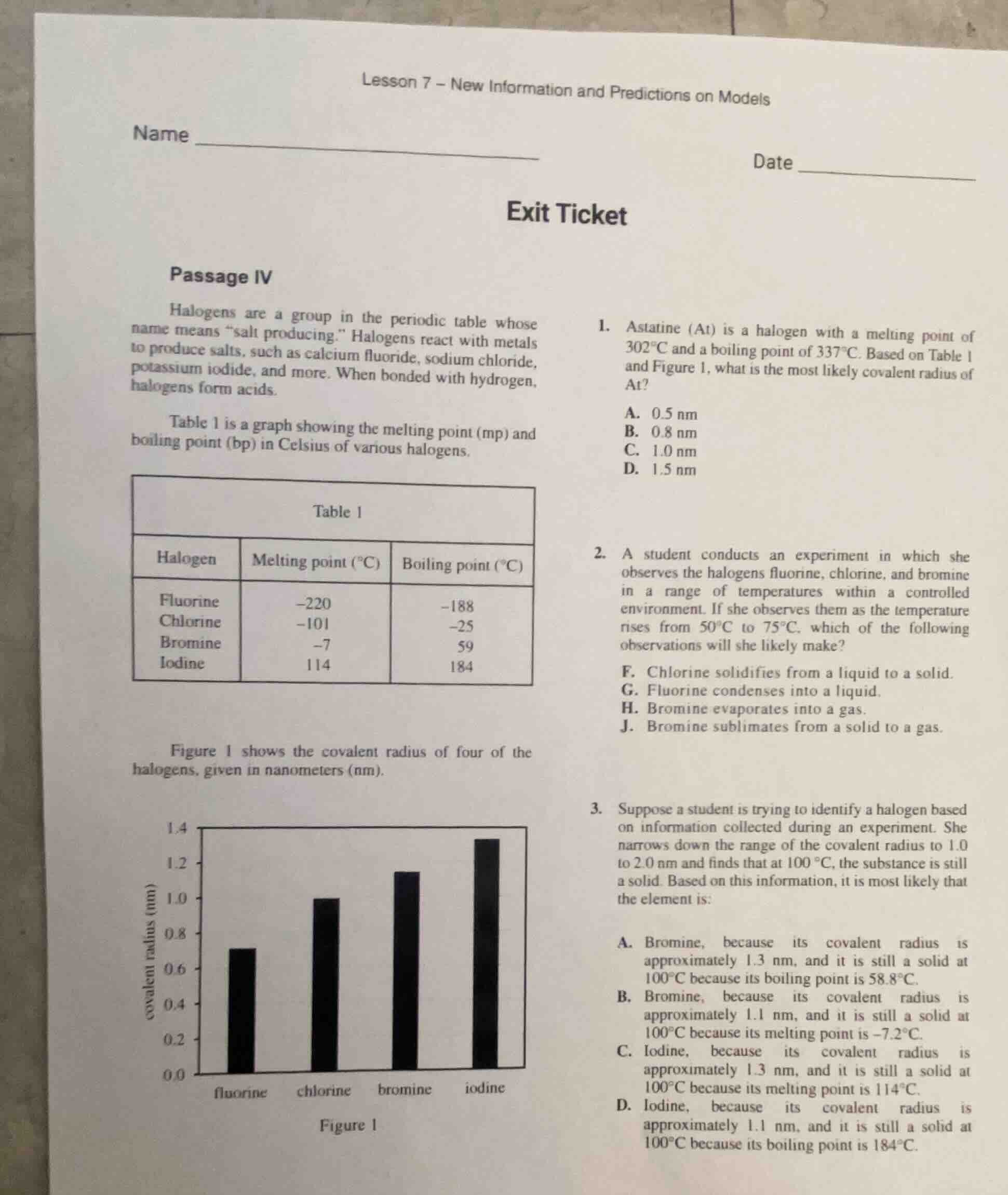
halogens are a group in the periodic table whose name means \salt producing.\ halogens react with metals to produce salts, such as calcium fluoride, sodium chloride, potassium iodide, and more. when bonded with hydrogen, halogens form acids.
table 1 is a graph showing the melting point (mp) and boiling point (bp) in celsius of various halogens.
table 1
| halogen | melting point (°c) | boiling point (°c) |
|---|---|---|
| chlorine | −101 | −25 |
| bromine | −7 | 59 |
| iodine | 114 | 184 |
figure 1 shows the covalent radius of four of the halogens, given in nanometers (nm).
figure 1
- astatine (at) is a halogen with a melting point of 302℃ and a boiling point of 337℃. based on table 1 and figure 1, what is the most likely covalent radius of at?
a. 0.5 nm
b. 0.8 nm
c. 1.0 nm
d. 1.5 nm
- a student conducts an experiment in which she observes the halogens fluorine, chlorine, and bromine in a range of temperatures within a controlled environment. if she observes them as the temperature rises from 50°c to 75°c, which of the following observations will she likely make?
f. chlorine solidifies from a liquid to a solid.
g. fluorine condenses into a liquid.
h. bromine evaporates into a gas.
j. bromine sublimates from a solid to a gas.
- suppose a student is trying to identify a halogen based on information collected during an experiment. she narrows down the range of the covalent radius to 1.0 to 2.0 nm and finds that at 100 °c, the substance is still a solid. based on this information, it is most likely that the element is:
a. bromine, because its covalent radius is approximately 1.3 nm, and it is still a solid at 100°c because its boiling point is 58.8°c.
b. bromine, because its covalent radius is approximately 1.1 nm, and it is still a solid at 100°c because its melting point is -7.2°c.
c. iodine, because its covalent radius is approximately 1.3 nm, and it is still a solid at 100°c because its melting point is 114°c.
d. iodine, because its covalent radius is approximately 1.1 nm, and it is still a solid at 100°c because its boiling point is 184°c.
Step1: Analyze halogen trend for Q1
From Figure 1, covalent radius increases down halogens: F (~0.7 nm) < Cl (~1.0 nm) < Br (~1.15 nm) < I (~1.3 nm). Astatine is below I, so radius >1.3 nm.
Step2: Analyze phase changes for Q2
Check Table 1: F (mp -220°C, bp -188°C) = gas at 50-75°C; Cl (mp -101°C, bp -25°C) = gas; Br (mp -7°C, bp 59°C). At 50-75°C, Br's bp is 59°C, so it evaporates to gas above 59°C.
Step3: Match data for Q3
From Figure 1, I has radius ~1.3 nm. Table 1: I mp=114°C, so at 100°C (below mp), it remains solid.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- D. 1.5 nm
- H. Bromine evaporates into a gas.
- C. Iodine, because its covalent radius is approximately 1.3 nm, and it is still a solid at 100°C because its melting point is 114°C.