QUESTION IMAGE
Question
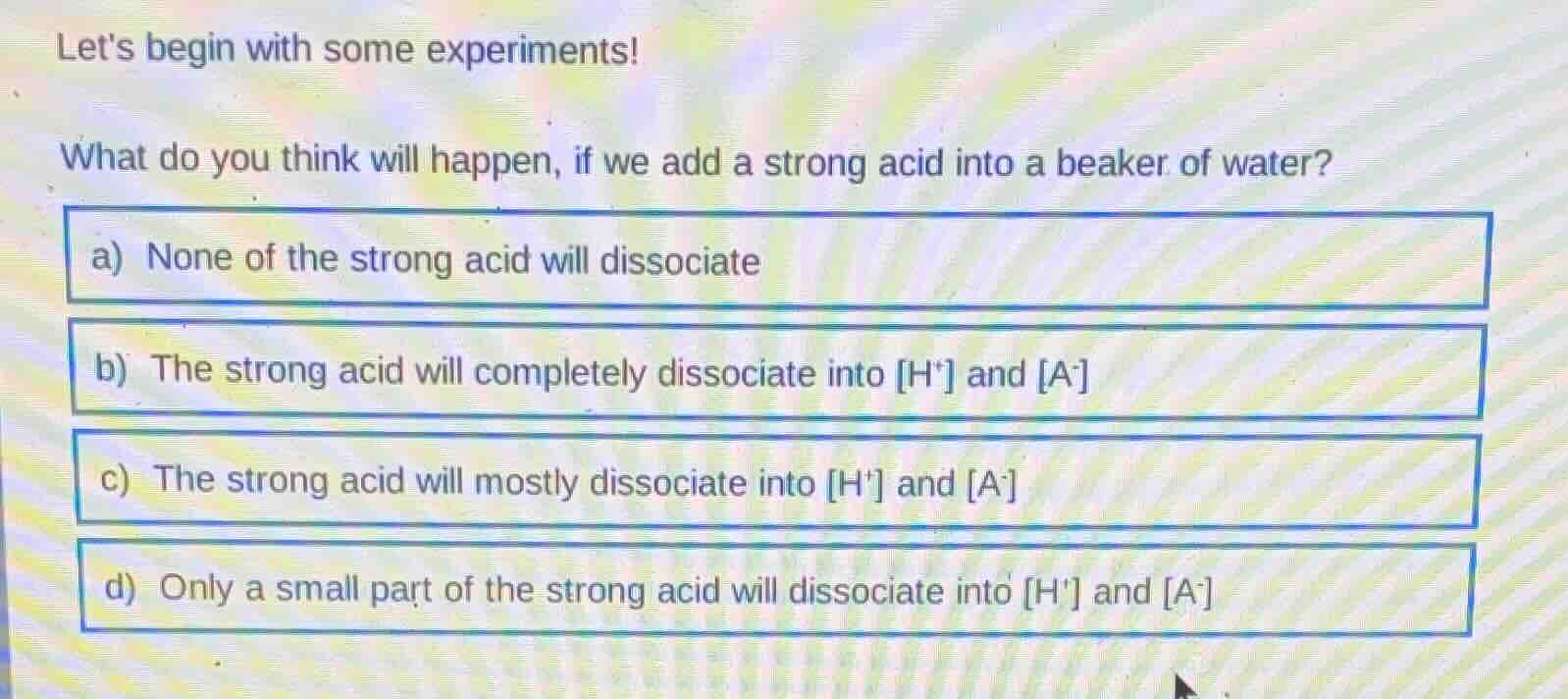
lets begin with some experiments!
what do you think will happen, if we add a strong acid into a beaker of water?
a) none of the strong acid will dissociate
b) the strong acid will completely dissociate into h⁺ and a⁻
c) the strong acid will mostly dissociate into h⁺ and a⁻
d) only a small part of the strong acid will dissociate into h⁺ and a⁻
By definition, strong acids are substances that fully break apart into hydrogen ions ($[H^+]$) and their conjugate base anions ($[A^-]$) when dissolved in water, with no measurable undissociated acid remaining.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
b) The strong acid will completely dissociate into $[H^+]$ and $[A^-]$