QUESTION IMAGE
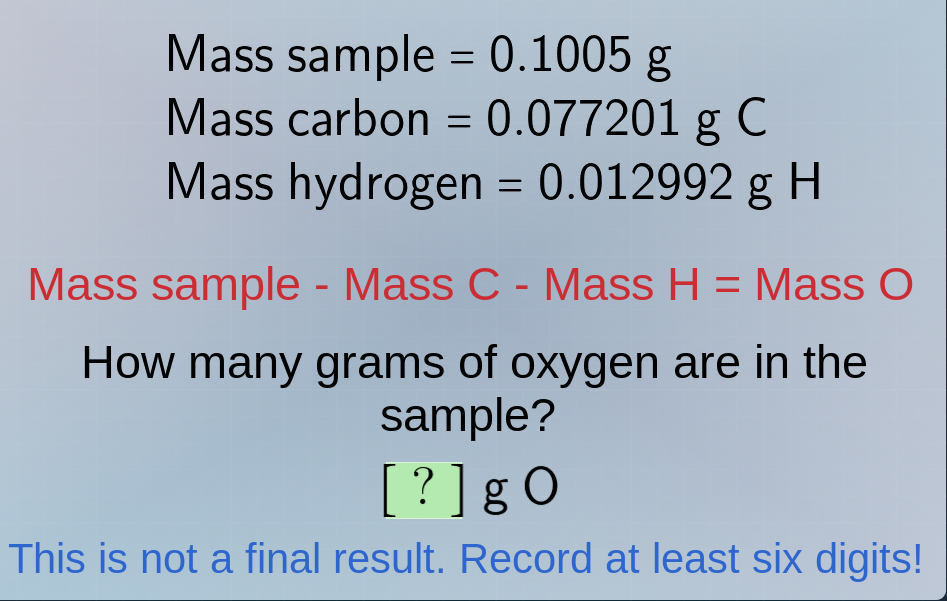
Question
mass sample = 0.1005 g
mass carbon = 0.077201 g c
mass hydrogen = 0.012992 g h
mass sample - mass c - mass h = mass o
how many grams of oxygen are in the sample?
? g o
this is not a final result. record at least six digits!
Step1: Identify the formula
The formula to find the mass of oxygen is \( \text{Mass O} = \text{Mass sample} - \text{Mass C} - \text{Mass H} \)
Step2: Substitute the values
We know that \( \text{Mass sample} = 0.1005 \, \text{g} \), \( \text{Mass C} = 0.077201 \, \text{g} \), and \( \text{Mass H} = 0.012992 \, \text{g} \). Substituting these values into the formula:
\( \text{Mass O} = 0.1005 - 0.077201 - 0.012992 \)
Step3: Perform the subtraction
First, subtract the mass of carbon from the mass of the sample: \( 0.1005 - 0.077201 = 0.023299 \)
Then, subtract the mass of hydrogen from the result: \( 0.023299 - 0.012992 = 0.010307 \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( 0.010307 \)