QUESTION IMAGE
Question
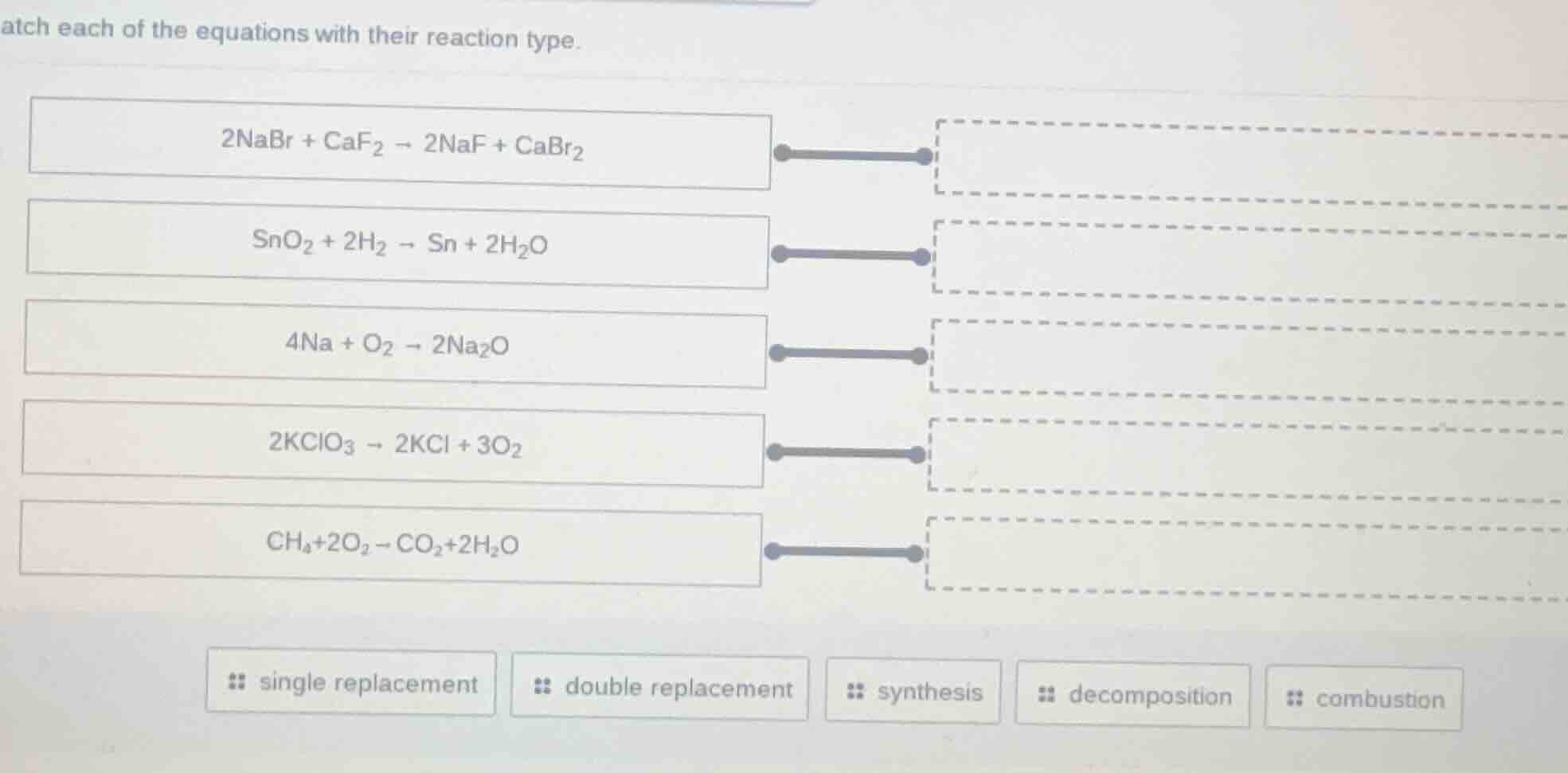
match each of the equations with their reaction type.
$2\text{nabr} + \text{caf}_2 \
ightarrow 2\text{naf} + \text{cabr}_2$
$\text{sno}_2 + 2\text{h}_2 \
ightarrow \text{sn} + 2\text{h}_2\text{o}$
$4\text{na} + \text{o}_2 \
ightarrow 2\text{na}_2\text{o}$
$2\text{kclo}_3 \
ightarrow 2\text{kcl} + 3\text{o}_2$
$\text{ch}_4+2\text{o}_2 \
ightarrow \text{co}_2+2\text{h}_2\text{o}$
single replacement double replacement synthesis decomposition combustion
- **$2\text{NaBr} + \text{CaF}_2
ightarrow 2\text{NaF} + \text{CaBr}_2$**: Ions ($\text{Na}^+/\text{Ca}^{2+}$ and $\text{Br}^-/F^-$) swap between two compounds, which defines a double replacement reaction.
- **$\text{SnO}_2 + 2\text{H}_2
ightarrow \text{Sn} + 2\text{H}_2\text{O}$**: A single element ($\text{H}_2$) replaces another element ($\text{Sn}$) in a compound, which defines a single replacement reaction.
- **$4\text{Na} + \text{O}_2
ightarrow 2\text{Na}_2\text{O}$**: Two reactants combine to form one single product, which defines a synthesis reaction.
- **$2\text{KClO}_3
ightarrow 2\text{KCl} + 3\text{O}_2$**: One reactant breaks down into multiple products, which defines a decomposition reaction.
- **$\text{CH}_4+2\text{O}_2
ightarrow \text{CO}_2+2\text{H}_2\text{O}$**: A hydrocarbon ($\text{CH}_4$) reacts with oxygen to produce carbon dioxide and water, which defines a combustion reaction.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $2\text{NaBr} + \text{CaF}_2
ightarrow 2\text{NaF} + \text{CaBr}_2$: double replacement
- $\text{SnO}_2 + 2\text{H}_2
ightarrow \text{Sn} + 2\text{H}_2\text{O}$: single replacement
- $4\text{Na} + \text{O}_2
ightarrow 2\text{Na}_2\text{O}$: synthesis
- $2\text{KClO}_3
ightarrow 2\text{KCl} + 3\text{O}_2$: decomposition
- $\text{CH}_4+2\text{O}_2
ightarrow \text{CO}_2+2\text{H}_2\text{O}$: combustion