QUESTION IMAGE
Question
missed this? watch kcv: names: molecular compounds, iwe: nomenclature: using the nomenclature flowchart: read section 5.8. you can click on the review to
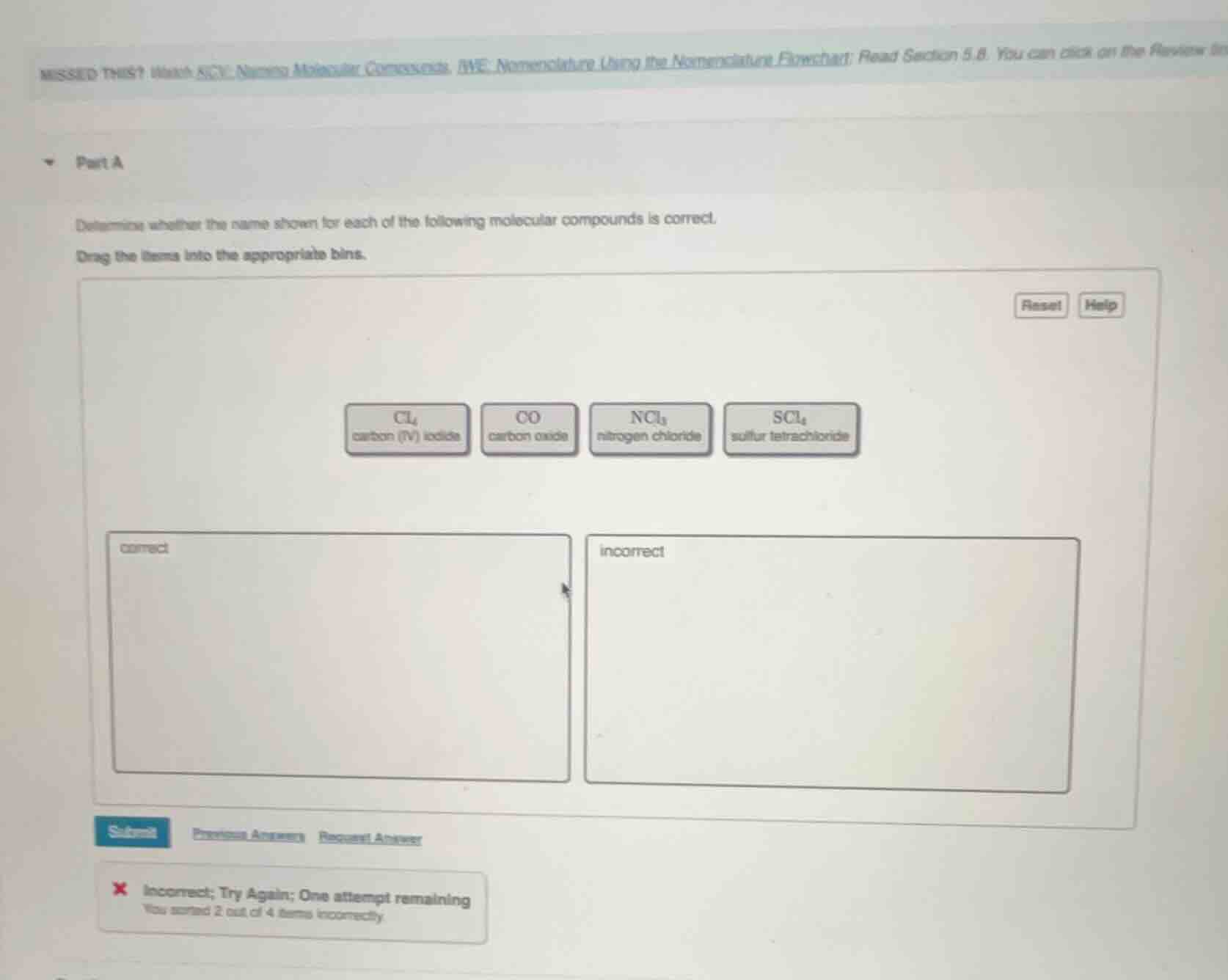
part a
determine whether the name shown for each of the following molecular compounds is correct.
drag the items into the appropriate bins.
ccl₄
carbon (iv) iodide
co
carbon oxide
ncl₃
nitrogen chloride
scl₄
sulfur tetrachloride
correct
incorrect
submit previous answers request answer
incorrect; try again; one attempt remaining
you sorted 2 out of 4 items incorrectly
Step1: Analyze \( CCl_4 \)
The formula \( CCl_4 \) is carbon tetrachloride. The given name is "carbon (IV) iodide" (wrong element, I vs Cl, and wrong prefix). So it's incorrect.
Step2: Analyze \( CO \)
\( CO \) is carbon monoxide. The given name is "carbon oxide" (missing the prefix "mono-"). So it's incorrect.
Step3: Analyze \( NCl_3 \)
\( NCl_3 \) is nitrogen trichloride (needs the prefix "tri-"). The given name is "nitrogen chloride" (missing prefix). So it's incorrect.
Step4: Analyze \( SCl_4 \)
\( SCl_4 \) is sulfur tetrachloride. The given name matches. So it's correct.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
correct: \( \boldsymbol{SCl_4} \) (sulfur tetrachloride)
incorrect: \( \boldsymbol{CCl_4} \) (carbon (IV) iodide), \( \boldsymbol{CO} \) (carbon oxide), \( \boldsymbol{NCl_3} \) (nitrogen chloride)