QUESTION IMAGE
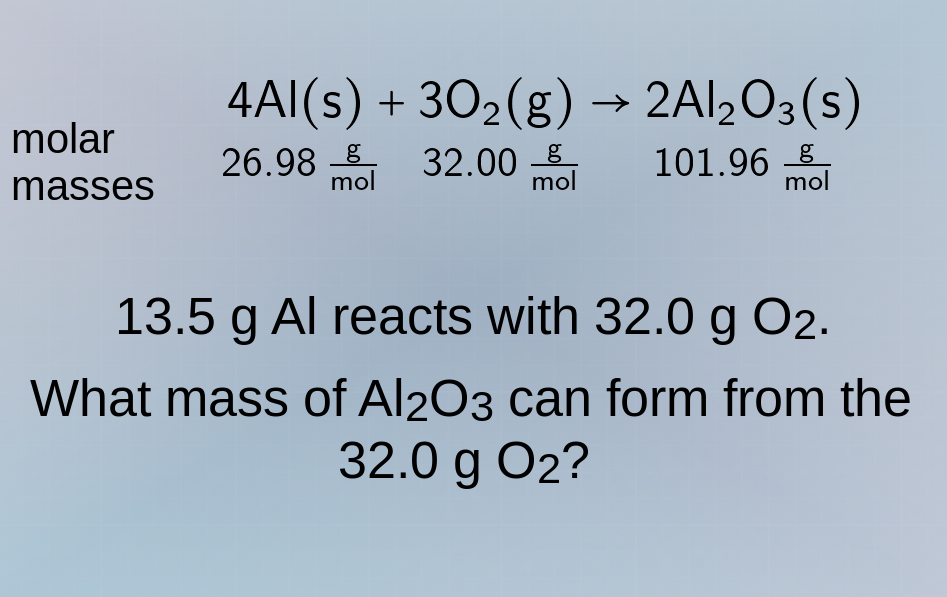
Question
molar masses
4al(s) + 3o₂(g) → 2al₂o₃(s)
26.98 g/mol 32.00 g/mol 101.96 g/mol
13.5 g al reacts with 32.0 g o₂.
what mass of al₂o₃ can form from the
32.0 g o₂?
Step1: Calculate moles of O₂
Molar mass of O₂ is \(32.00 \frac{g}{mol}\). Moles of O₂ = \(\frac{mass}{molar\ mass}\) = \(\frac{32.0\ g}{32.00\ \frac{g}{mol}} = 1\ mol\).
Step2: Use stoichiometry to find moles of Al₂O₃
From the reaction \(4Al(s)+3O_2(g)
ightarrow2Al_2O_3(s)\), the mole ratio of \(O_2\) to \(Al_2O_3\) is \(3:2\). So moles of \(Al_2O_3\) = \(\frac{2}{3}\times\) moles of \(O_2\) = \(\frac{2}{3}\times1\ mol=\frac{2}{3}\ mol\).
Step3: Calculate mass of Al₂O₃
Molar mass of \(Al_2O_3\) is \(101.96\ \frac{g}{mol}\). Mass = moles \(\times\) molar mass = \(\frac{2}{3}\ mol\times101.96\ \frac{g}{mol}\approx67.97\ g\).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(\approx 68.0\ g\) (or more precisely \(67.97\ g\))