QUESTION IMAGE
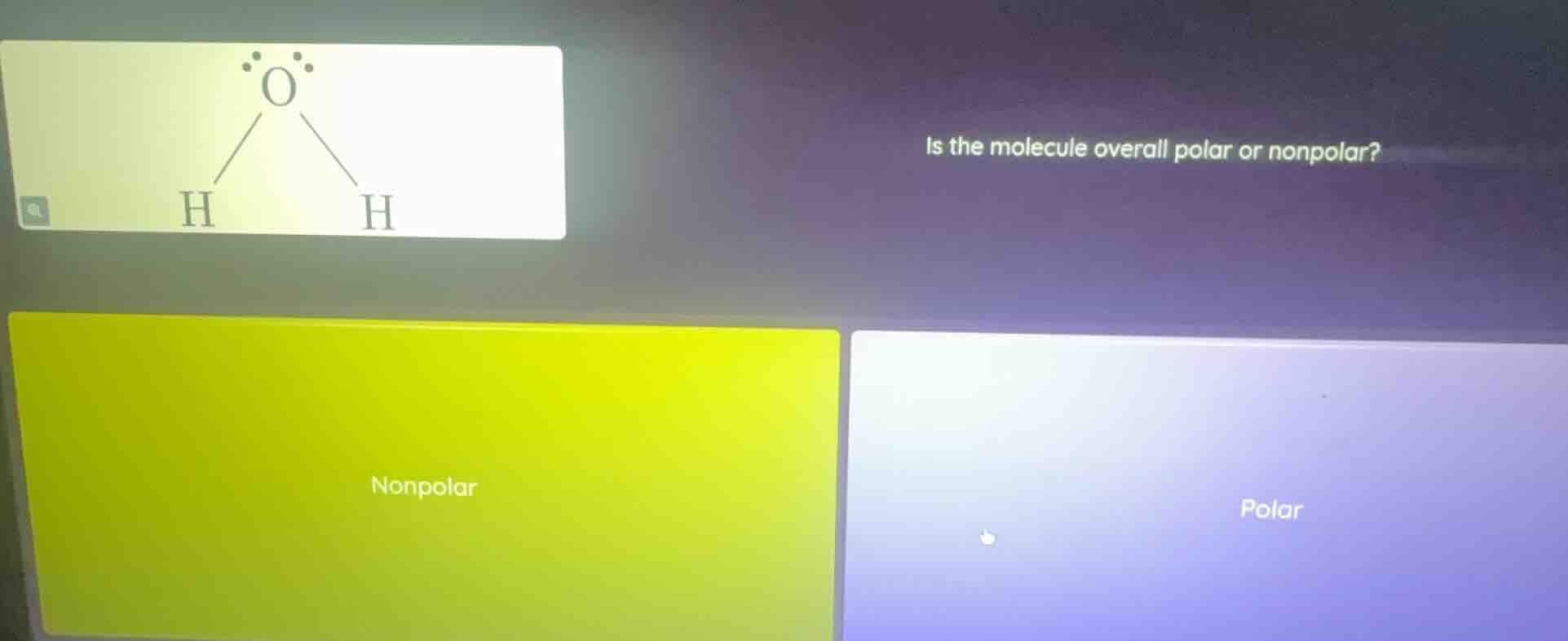
Question
is the molecule overall polar or nonpolar?
options: nonpolar, polar
Brief Explanations
The molecule shown is \( \ce{H_2O} \) (water). The oxygen atom is more electronegative than hydrogen, creating polar \( \ce{O - H} \) bonds. The molecular geometry of water is bent (due to two lone pairs on oxygen), so the dipole moments of the two \( \ce{O - H} \) bonds do not cancel out. This results in a net dipole moment, making the water molecule polar.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Polar