QUESTION IMAGE
Question
multiple-choice question
how do you determine the number of neutrons?
protons - atomic mass
mass # - atomic number
protons + electrons
rewatch
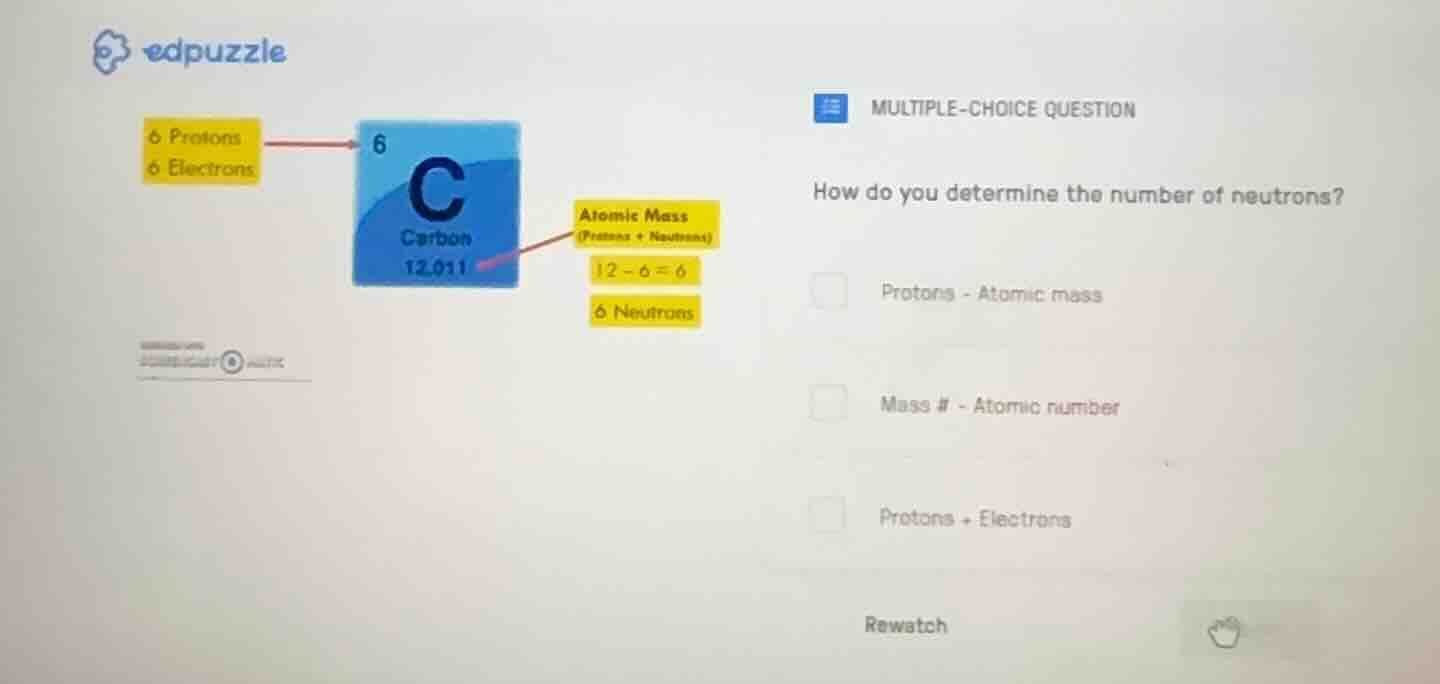
6 protons
6 electrons
atomic mass (protons + neutrons)
12 - 6 = 6
6 neutrons
c
carbon
12.011
Brief Explanations
The atomic mass (mass number) is the sum of protons and neutrons. The atomic number equals the number of protons. Rearranging gives neutrons = mass number - atomic number, which matches the carbon example: $12 - 6 = 6$ neutrons.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Mass # - Atomic number