QUESTION IMAGE
Question
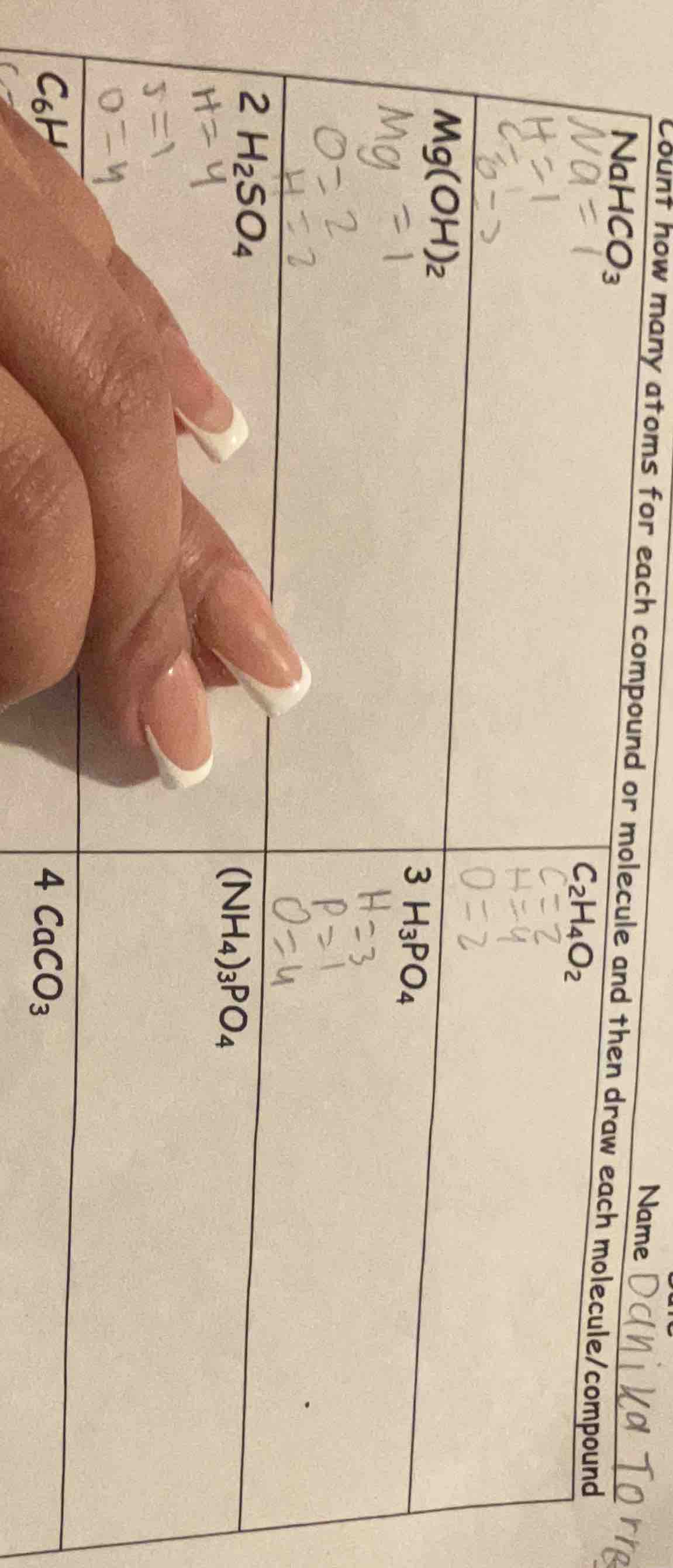
name: daniela torres
count how many atoms for each compound or molecule and then draw each molecule/compound
$ce{nahco3}$
$ce{c2h4o2}$
$ce{mg(oh)2}$
$ce{3 h3po4}$
$ce{2 h2so4}$
$ce{(nh4)3po4}$
$ce{c6h}$
$ce{4 caco3}$
Step1: Count atoms in $\text{NaHCO}_3$
$\text{Na}=1$, $\text{H}=1$, $\text{C}=1$, $\text{O}=3$
Step2: Count atoms in $\text{Mg(OH)}_2$
$\text{Mg}=1$, $\text{O}=2$, $\text{H}=2$
Step3: Count atoms in $2\text{H}_2\text{SO}_4$
$\text{H}=2\times2=4$, $\text{S}=2\times1=2$, $\text{O}=2\times4=8$
Step4: Count atoms in $\text{C}_2\text{H}_4\text{O}_2$
$\text{C}=2$, $\text{H}=4$, $\text{O}=2$
Step5: Count atoms in $3\text{H}_3\text{PO}_4$
$\text{H}=3\times3=9$, $\text{P}=3\times1=3$, $\text{O}=3\times4=12$
Step6: Count atoms in $(\text{NH}_4)_3\text{PO}_4$
$\text{N}=3$, $\text{H}=3\times4=12$, $\text{P}=1$, $\text{O}=4$
Step7: Count atoms in $4\text{CaCO}_3$
$\text{Ca}=4\times1=4$, $\text{C}=4\times1=4$, $\text{O}=4\times3=12$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $\text{NaHCO}_3$: 1 Na, 1 H, 1 C, 3 O
- $\text{Mg(OH)}_2$: 1 Mg, 2 O, 2 H
- $2\text{H}_2\text{SO}_4$: 4 H, 2 S, 8 O
- $\text{C}_2\text{H}_4\text{O}_2$: 2 C, 4 H, 2 O
- $3\text{H}_3\text{PO}_4$: 9 H, 3 P, 12 O
- $(\text{NH}_4)_3\text{PO}_4$: 3 N, 12 H, 1 P, 4 O
- $4\text{CaCO}_3$: 4 Ca, 4 C, 12 O
(Note: Structural drawing instructions are omitted as they require visual representation; atom counts are completed as requested)