QUESTION IMAGE
Question
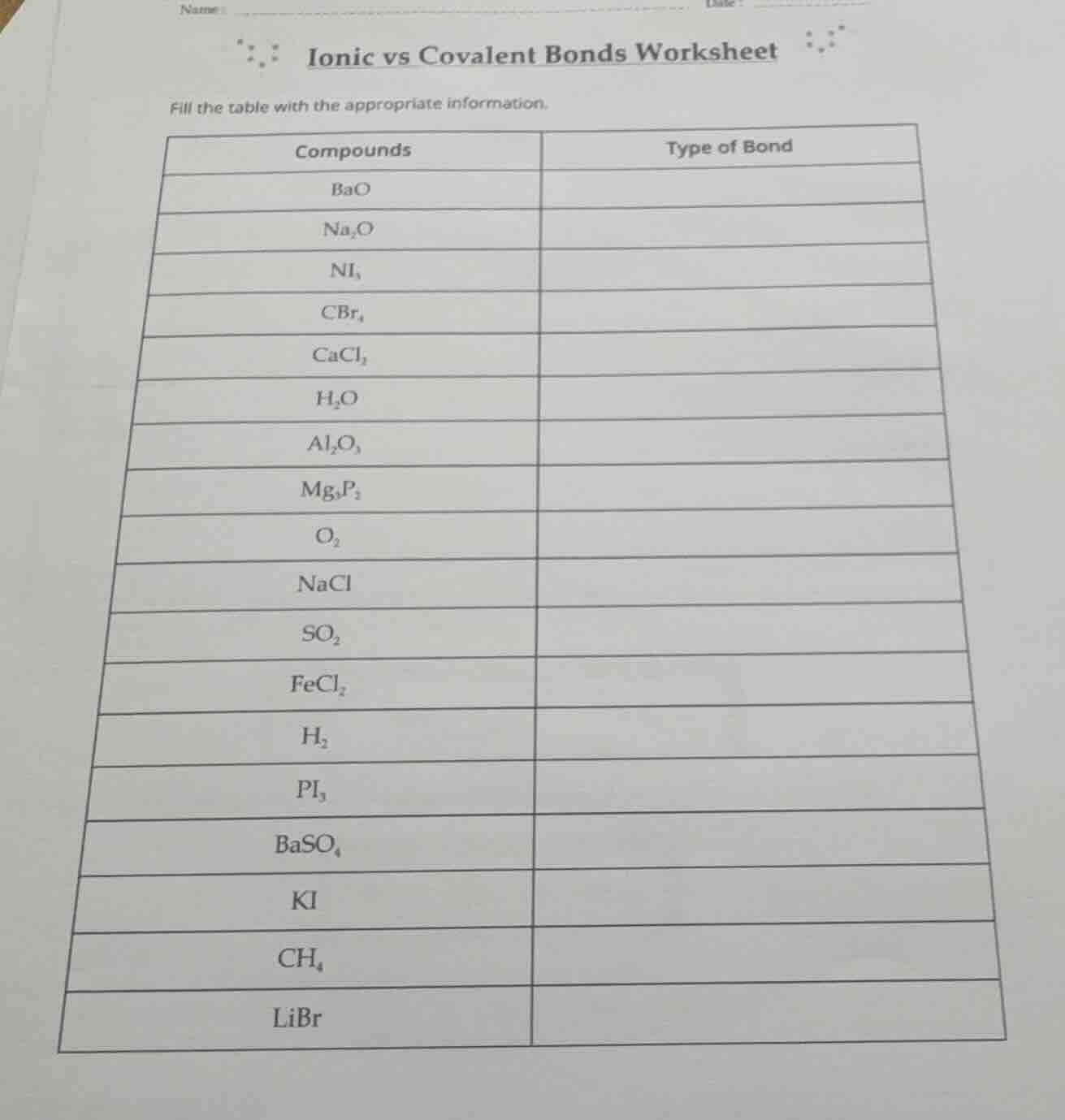
name
date
ionic vs covalent bonds worksheet
fill the table with the appropriate information.
compounds | type of bond
bao |
na₂o |
ni₃ |
cbr₄ |
cacl₂ |
h₂o |
al₂o₃ |
mg₃p₂ |
o₂ |
nacl |
so₂ |
fecl₂ |
h₂ |
pi₃ |
baso₄ |
ki |
ch₄ |
libr |
Step1: Classify ionic/covalent bonds
Ionic bonds form between metals (groups 1-2, transition metals) and nonmetals; covalent bonds form between nonmetals (including same-element molecules). For ionic compounds with polyatomic ions, the overall compound is ionic.
Step2: Assign bond type to each compound
- $\text{BaO}$: Ba (metal) + O (nonmetal) → Ionic
- $\text{Na}_2\text{O}$: Na (metal) + O (nonmetal) → Ionic
- $\text{NI}_3$: N (nonmetal) + I (nonmetal) → Covalent
- $\text{CBr}_4$: C (nonmetal) + Br (nonmetal) → Covalent
- $\text{CaCl}_2$: Ca (metal) + Cl (nonmetal) → Ionic
- $\text{H}_2\text{O}$: H (nonmetal) + O (nonmetal) → Covalent
- $\text{Al}_2\text{O}_3$: Al (metal) + O (nonmetal) → Ionic
- $\text{Mg}_3\text{P}_2$: Mg (metal) + P (nonmetal) → Ionic
- $\text{O}_2$: Same nonmetal atoms → Covalent
- $\text{NaCl}$: Na (metal) + Cl (nonmetal) → Ionic
- $\text{SO}_2$: S (nonmetal) + O (nonmetal) → Covalent
- $\text{FeCl}_2$: Fe (metal) + Cl (nonmetal) → Ionic
- $\text{H}_2$: Same nonmetal atoms → Covalent
- $\text{PI}_3$: P (nonmetal) + I (nonmetal) → Covalent
- $\text{BaSO}_4$: Ba (metal) + $\text{SO}_4^{2-}$ (polyatomic ion) → Ionic
- $\text{KI}$: K (metal) + I (nonmetal) → Ionic
- $\text{CH}_4$: C (nonmetal) + H (nonmetal) → Covalent
- $\text{LiBr}$: Li (metal) + Br (nonmetal) → Ionic
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Compounds | Type of Bond |
|---|---|
| $\text{Na}_2\text{O}$ | Ionic |
| $\text{NI}_3$ | Covalent |
| $\text{CBr}_4$ | Covalent |
| $\text{CaCl}_2$ | Ionic |
| $\text{H}_2\text{O}$ | Covalent |
| $\text{Al}_2\text{O}_3$ | Ionic |
| $\text{Mg}_3\text{P}_2$ | Ionic |
| $\text{O}_2$ | Covalent |
| $\text{NaCl}$ | Ionic |
| $\text{SO}_2$ | Covalent |
| $\text{FeCl}_2$ | Ionic |
| $\text{H}_2$ | Covalent |
| $\text{PI}_3$ | Covalent |
| $\text{BaSO}_4$ | Ionic |
| $\text{KI}$ | Ionic |
| $\text{CH}_4$ | Covalent |
| $\text{LiBr}$ | Ionic |