QUESTION IMAGE
Question
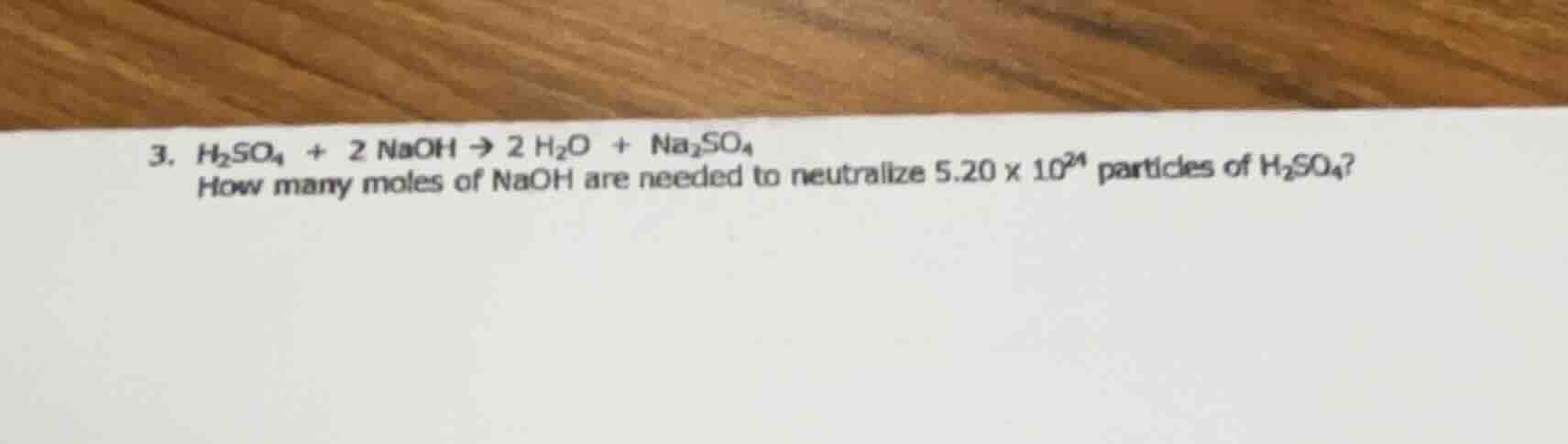
- $\ce{h_{2}so_{4} + 2 naoh \
ightarrow 2 h_{2}o + na_{2}so_{4}}$ how many moles of naoh are needed to neutralize $5.20 \times 10^{24}$ particles of $\ce{h_{2}so_{4}}$?
Step1: Find moles of \( H_2SO_4 \)
Using Avogadro's number (\( N_A = 6.022\times10^{23} \) particles/mol), moles of \( H_2SO_4 \) (\( n_{H_2SO_4} \)) is \( n = \frac{N}{N_A} \).
\( n_{H_2SO_4} = \frac{5.20\times10^{24}}{6.022\times10^{23}} \approx 8.635 \) mol.
Step2: Use stoichiometry from reaction
Reaction: \( H_2SO_4 + 2NaOH
ightarrow 2H_2O + Na_2SO_4 \).
Mole ratio \( \frac{n_{NaOH}}{n_{H_2SO_4}} = \frac{2}{1} \), so \( n_{NaOH} = 2 \times n_{H_2SO_4} \).
Step3: Calculate moles of \( NaOH \)
Substitute \( n_{H_2SO_4} \approx 8.635 \) mol:
\( n_{NaOH} = 2 \times 8.635 \approx 17.27 \) mol (or more precisely, using exact fraction: \( \frac{5.20\times10^{24}}{6.022\times10^{23}} \times 2 \approx \frac{5.20\times2\times10^{24}}{6.022\times10^{23}} = \frac{10.4\times10^{24}}{6.022\times10^{23}} \approx 17.3 \) mol).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Approximately \( \boldsymbol{17.3} \) moles (or more precisely ~17.27 mol) of \( NaOH \) are needed.