QUESTION IMAGE
Question
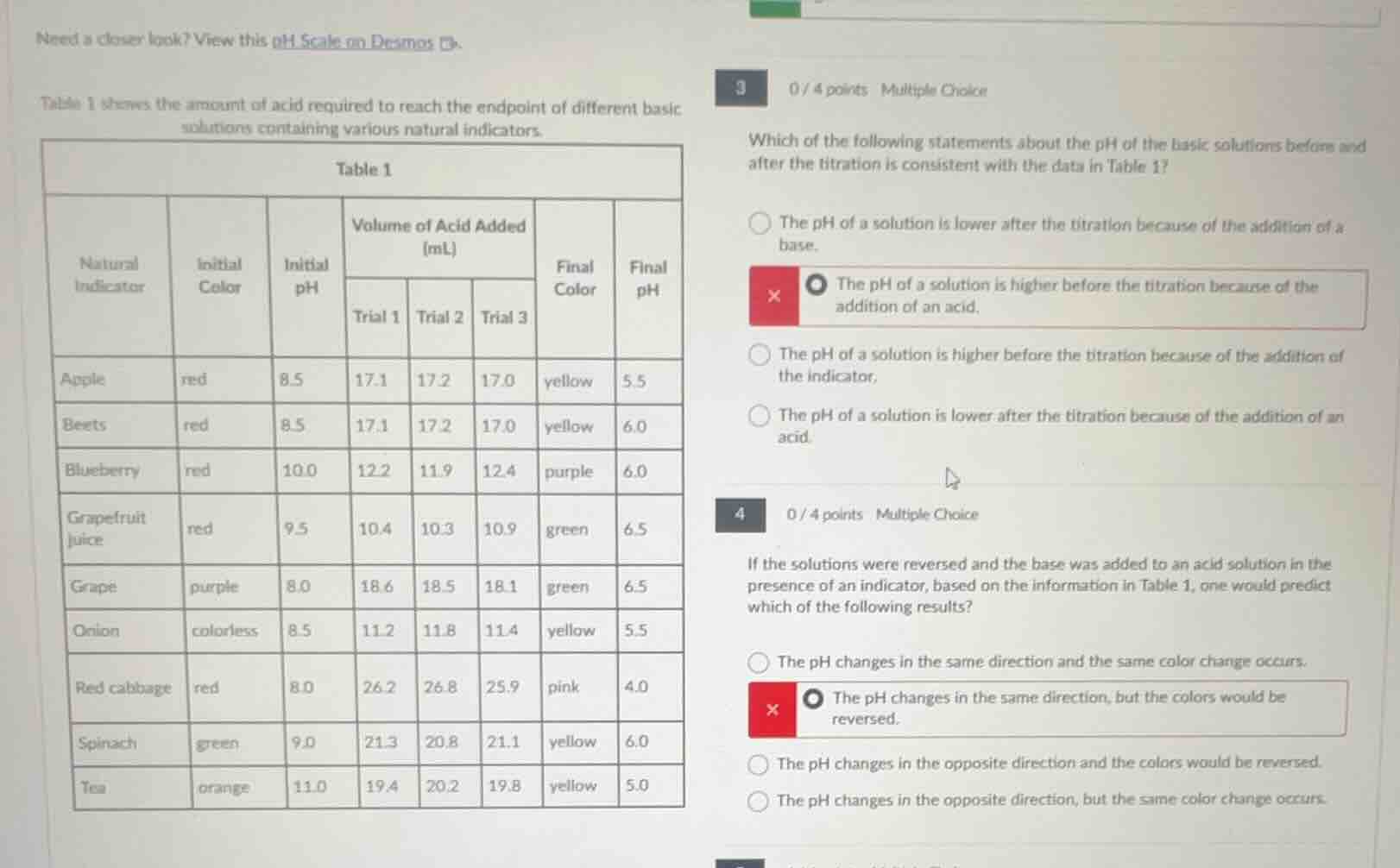
need a closer look? view this ph scale on desmos.table 1 shows the amount of acid required to reach the endpoint of different basic solutions containing various natural indicators.table 1| natural indicator | initial color | initial ph | volume of acid added (ml) | | | final color | final ph || ---- | ---- | ---- | ---- | ---- | ---- | ---- | ---- || | | | trial 1 | trial 2 | trial 3 | | || apple | red | 8.5 | 17.1 | 17.2 | 17.0 | yellow | 5.5 || beets | red | 8.5 | 17.1 | 17.2 | 17.0 | yellow | 6.0 || blueberry | red | 10.0 | 12.2 | 11.9 | 12.4 | purple | 6.0 || grapefruit juice | red | 9.5 | 10.4 | 10.3 | 10.9 | green | 6.5 || grape | purple | 8.0 | 18.6 | 18.5 | 18.1 | green | 6.5 || onion | colorless | 8.5 | 11.2 | 11.8 | 11.4 | yellow | 5.5 || red cabbage | red | 8.0 | 26.2 | 26.8 | 25.9 | pink | 4.0 || spinach | green | 9.0 | 21.3 | 20.8 | 21.1 | yellow | 6.0 || tea | orange | 11.0 | 19.4 | 20.2 | 19.8 | yellow | 5.0 |3 0 / 4 points multiple choicewhich of the following statements about the ph of the basic solutions before and after the titration is consistent with the data in table 1?- the ph of a solution is lower after the titration because of the addition of a base.- the ph of a solution is higher before the titration because of the addition of an acid.- the ph of a solution is higher before the titration because of the addition of the indicator.- the ph of a solution is lower after the titration because of the addition of an acid.4 0 / 4 points multiple choiceif the solutions were reversed and the base was added to an acid solution in the presence of an indicator, based on the information in table 1, one would predict which of the following results?- the ph changes in the same direction and the same color change occurs.- the ph changes in the same direction, but the colors would be reversed.- the ph changes in the opposite direction and the colors would be reversed.- the ph changes in the opposite direction, but the same color change occurs.
For question 3:
- Analyze Table 1: All initial pH values are >7 (basic, higher pH), final pH values are <7 or neutral (lower pH). Titration adds acid to base, so acid lowers pH.
- Eliminate incorrect options:
- Option 1: Wrong reason (says base added, not acid).
- Option 2: Wrong reason (initial high pH is due to being basic, not acid addition).
- Option 3: Wrong reason (indicator doesn't cause high initial pH; solution is basic initially).
- Option 4: Correct: Acid addition lowers pH, matching initial (high) vs final (low) pH data.
For question 4:
- Original titration: Acid added to base → pH decreases (from high to low), color changes from initial to final.
- Reversed titration: Base added to acid → pH increases (from low to high), which is the opposite direction. Natural indicators change color based on pH range, so the color sequence would reverse (e.g., original final color becomes initial, original initial becomes final).
- Eliminate incorrect options:
- Option 1: pH direction is opposite, not same.
- Option 2: pH direction is opposite, not same.
- Option 3: Correct: pH changes opposite direction, colors reverse.
- Option 4: Color change would reverse, not stay same.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- D. The pH of a solution is lower after the titration because of the addition of an acid.
- C. The pH changes in the opposite direction and the colors would be reversed.