QUESTION IMAGE
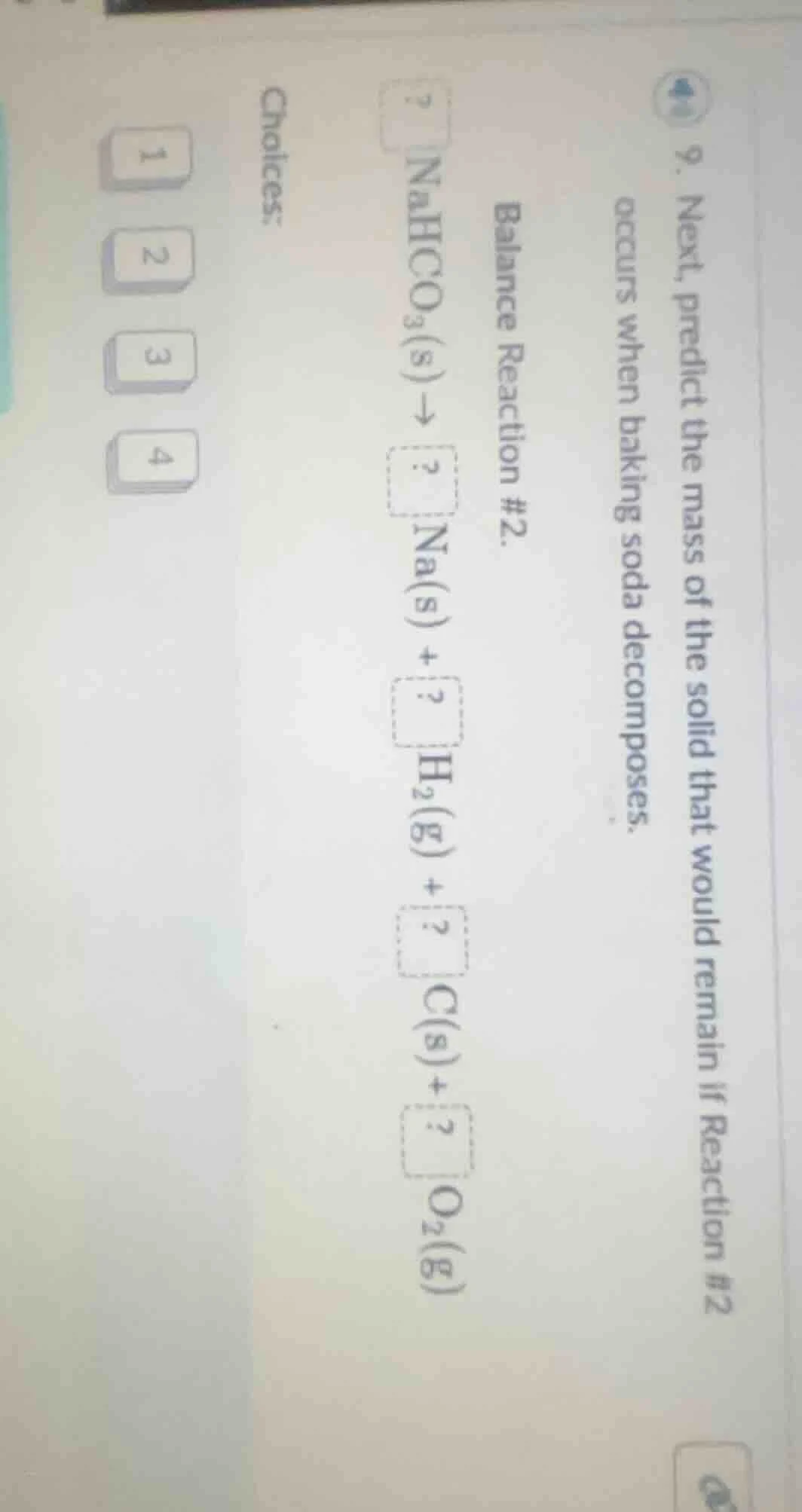
Question
- next, predict the mass of the solid that would remain if reaction #2 occurs when baking soda decomposes.
balance reaction #2.
$\boldsymbol{?}\\ \ce{nahco_{3}(s) \
ightarrow \boldsymbol{?}\\ \ce{na(s)} + \boldsymbol{?}\\ \ce{h_{2}(g)} + \boldsymbol{?}\\ \ce{c(s)} + \boldsymbol{?}\\ \ce{o_{2}(g)}}$
choices:
1 2 3 4
Step1: Balance Na atoms
Step2: Balance H atoms
H is already balanced (2 on each side).
Step3: Balance C atoms
C is already balanced (1 on each side).
Step4: Balance O atoms
Adjust O₂ coefficient:
Verify all atoms:
- Na: 2 on both sides
- H: 2 on both sides
- C: 2 on both sides
- O: 6 on both sides
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The coefficients (from left to right) are 2, 2, 1, 2, 3. The solid products are $\text{Na}(s)$ and $\text{C}(s)$; their total molar mass per 2 moles of $\text{NaHCO}_3$ is $(2\times22.99) + (2\times12.01) = 70.00$ g. For 1 mole of $\text{NaHCO}_3$ (84.01 g), the remaining solid mass is 35.00 g.