QUESTION IMAGE
Question
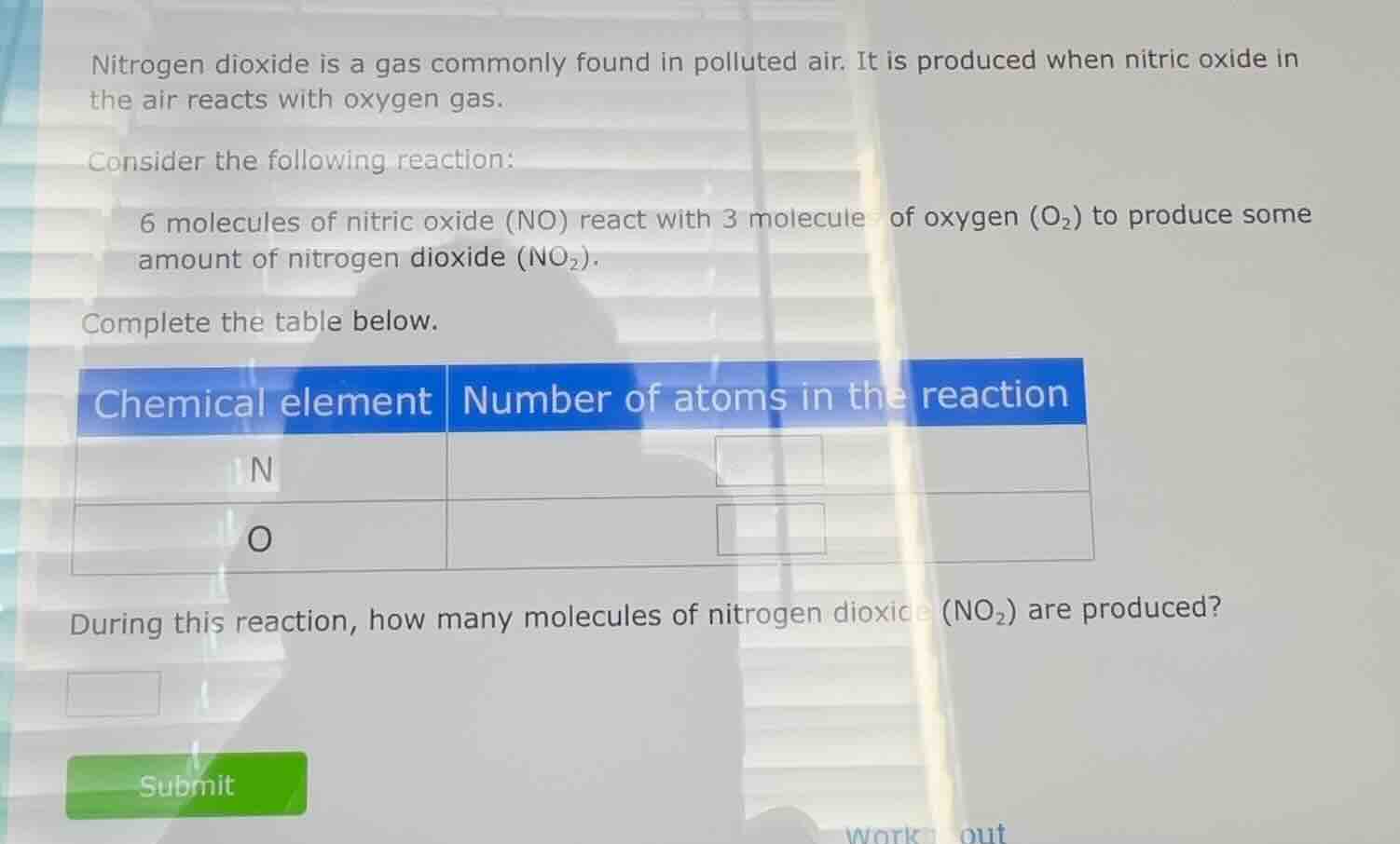
nitrogen dioxide is a gas commonly found in polluted air. it is produced when nitric oxide in the air reacts with oxygen gas. consider the following reaction: 6 molecules of nitric oxide (no) react with 3 molecules of oxygen (o₂) to produce some amount of nitrogen dioxide (no₂). complete the table below. chemical element | number of atoms in the reaction n | o | during this reaction, how many molecules of nitrogen dioxide (no₂) are produced? submit work out
For the number of N atoms:
Step1: Analyze NO molecules
Each NO molecule has 1 N atom. There are 6 NO molecules. So, number of N atoms = 6 × 1 = 6.
Step2: Confirm conservation
In a chemical reaction, the number of atoms of each element is conserved. So, N atoms in products (NO₂) will also be 6.
Step1: Calculate O from NO
Each NO has 1 O atom. 6 NO molecules have 6 × 1 = 6 O atoms.
Step2: Calculate O from O₂
Each O₂ has 2 O atoms. 3 O₂ molecules have 3 × 2 = 6 O atoms.
Step3: Total O atoms
Total O atoms = 6 (from NO) + 6 (from O₂) = 12.
Step1: Balance the reaction
The reaction is \( 2NO + O_2
ightarrow 2NO_2 \) (simplified from the given 6 NO and 3 O₂: divide by 3, we get 2 NO + 1 O₂ → 2 NO₂).
Step2: Use the ratio
Given 6 NO (which is 3 times 2 NO) and 3 O₂ (3 times 1 O₂), the amount of NO₂ produced will be 3 times 2 NO₂, so 6 NO₂ molecules.
Or, using atom conservation: Each NO₂ has 1 N and 2 O. We have 6 N atoms, so number of NO₂ molecules from N: 6 (since 1 N per NO₂). Check O: 6 NO₂ have 6 × 2 = 12 O atoms, which matches the total O atoms calculated earlier. So, number of NO₂ molecules is 6.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
(for N atoms): 6