QUESTION IMAGE
Question
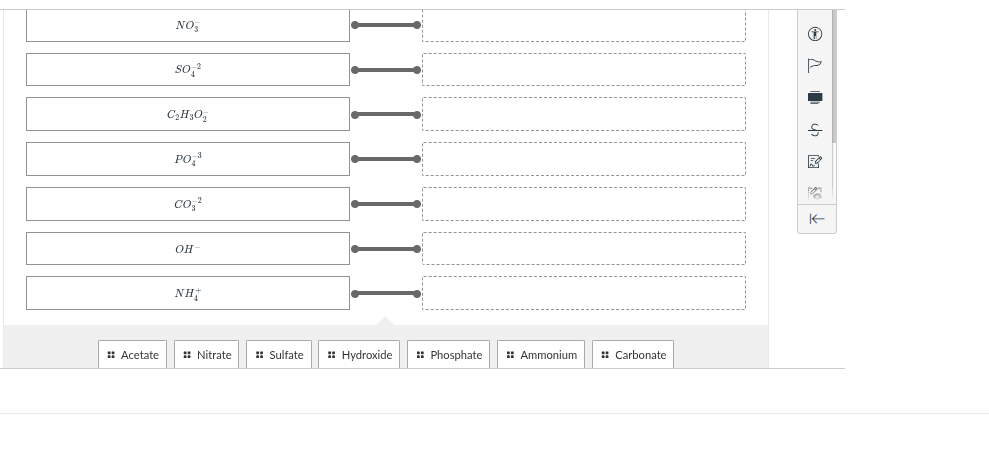
no₃⁻ so₄²⁻ c₂h₃o₂⁻ po₄³⁻ co₃²⁻ oh⁻ nh₄⁺ acetate nitrate sulfate hydroxide phosphate ammonium carbonate
To solve this ion - name matching problem, we analyze each ion and its corresponding name:
Step 1: Analyze \(NO_3^-\)
The ion \(NO_3^-\) is the nitrate ion. So we match \(NO_3^-\) with "Nitrate".
Step 2: Analyze \(SO_4^{2 -}\)
The ion \(SO_4^{2 -}\) is the sulfate ion. So we match \(SO_4^{2 -}\) with "Sulfate".
Step 3: Analyze \(C_2H_3O_2^-\)
The ion \(C_2H_3O_2^-\) is the acetate ion. So we match \(C_2H_3O_2^-\) with "Acetate".
Step 4: Analyze \(PO_4^{3 -}\)
The ion \(PO_4^{3 -}\) is the phosphate ion. So we match \(PO_4^{3 -}\) with "Phosphate".
Step 5: Analyze \(CO_3^{2 -}\)
The ion \(CO_3^{2 -}\) is the carbonate ion. So we match \(CO_3^{2 -}\) with "Carbonate".
Step 6: Analyze \(OH^-\)
The ion \(OH^-\) is the hydroxide ion. So we match \(OH^-\) with "Hydroxide".
Step 7: Analyze \(NH_4^+\)
The ion \(NH_4^+\) is the ammonium ion. So we match \(NH_4^+\) with "Ammonium".
Final Matching:
- \(NO_3^-\) \(\longleftrightarrow\) Nitrate
- \(SO_4^{2 -}\) \(\longleftrightarrow\) Sulfate
- \(C_2H_3O_2^-\) \(\longleftrightarrow\) Acetate
- \(PO_4^{3 -}\) \(\longleftrightarrow\) Phosphate
- \(CO_3^{2 -}\) \(\longleftrightarrow\) Carbonate
- \(OH^-\) \(\longleftrightarrow\) Hydroxide
- \(NH_4^+\) \(\longleftrightarrow\) Ammonium
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve this ion - name matching problem, we analyze each ion and its corresponding name:
Step 1: Analyze \(NO_3^-\)
The ion \(NO_3^-\) is the nitrate ion. So we match \(NO_3^-\) with "Nitrate".
Step 2: Analyze \(SO_4^{2 -}\)
The ion \(SO_4^{2 -}\) is the sulfate ion. So we match \(SO_4^{2 -}\) with "Sulfate".
Step 3: Analyze \(C_2H_3O_2^-\)
The ion \(C_2H_3O_2^-\) is the acetate ion. So we match \(C_2H_3O_2^-\) with "Acetate".
Step 4: Analyze \(PO_4^{3 -}\)
The ion \(PO_4^{3 -}\) is the phosphate ion. So we match \(PO_4^{3 -}\) with "Phosphate".
Step 5: Analyze \(CO_3^{2 -}\)
The ion \(CO_3^{2 -}\) is the carbonate ion. So we match \(CO_3^{2 -}\) with "Carbonate".
Step 6: Analyze \(OH^-\)
The ion \(OH^-\) is the hydroxide ion. So we match \(OH^-\) with "Hydroxide".
Step 7: Analyze \(NH_4^+\)
The ion \(NH_4^+\) is the ammonium ion. So we match \(NH_4^+\) with "Ammonium".
Final Matching:
- \(NO_3^-\) \(\longleftrightarrow\) Nitrate
- \(SO_4^{2 -}\) \(\longleftrightarrow\) Sulfate
- \(C_2H_3O_2^-\) \(\longleftrightarrow\) Acetate
- \(PO_4^{3 -}\) \(\longleftrightarrow\) Phosphate
- \(CO_3^{2 -}\) \(\longleftrightarrow\) Carbonate
- \(OH^-\) \(\longleftrightarrow\) Hydroxide
- \(NH_4^+\) \(\longleftrightarrow\) Ammonium