QUESTION IMAGE
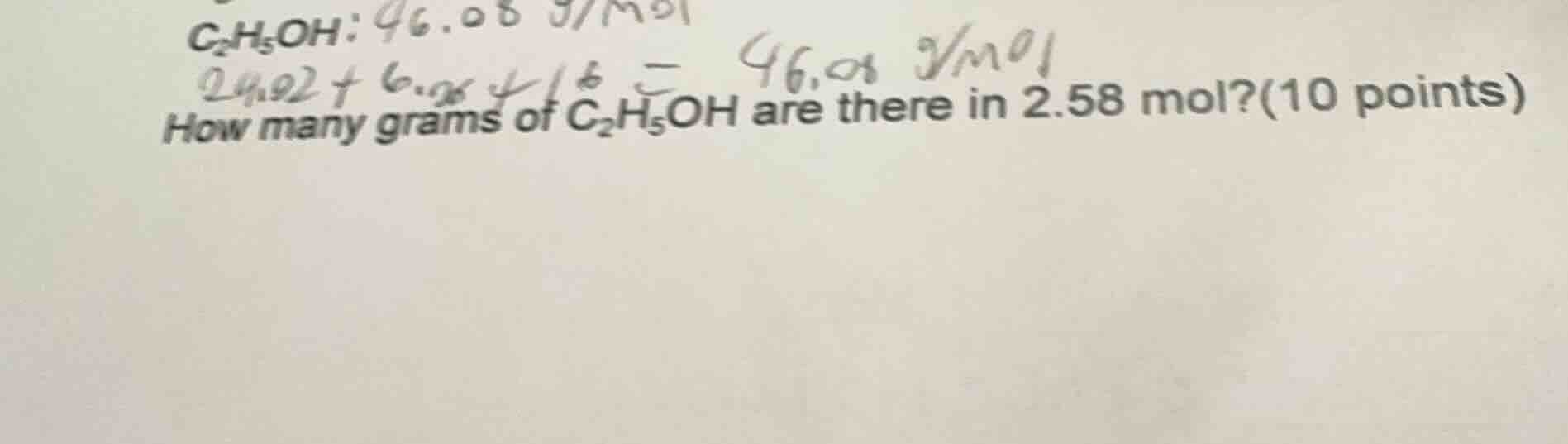
Question
c₂h₅oh: 46.08 g/mol
how many grams of c₂h₅oh are there in 2.58 mol?(10 points)
Step1: Recall the formula for mass calculation
The formula relating moles (\(n\)), molar mass (\(M\)), and mass (\(m\)) is \(m = n\times M\). Here, the number of moles \(n = 2.58\space mol\) and the molar mass \(M\) of \(\ce{C2H5OH}\) is given as \(46.08\space g/mol\).
Step2: Substitute the values into the formula
Substitute \(n = 2.58\space mol\) and \(M = 46.08\space g/mol\) into the formula \(m=n\times M\). So, \(m = 2.58\space mol\times46.08\space g/mol\).
Step3: Calculate the product
Calculate \(2.58\times46.08\). \(2.58\times46.08 = 2.58\times(46 + 0.08)=2.58\times46+2.58\times0.08 = 118.68+0.2064 = 118.8864\space g\).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The mass of \(\ce{C2H5OH}\) is \(\boldsymbol{118.89\space g}\) (rounded to two decimal places) or \(118.8864\space g\) (exact value from calculation).