QUESTION IMAGE
Question
organic chemistry problem set – unit assignment
you may refer to your course notes/lessons to answer the following. submit to d2l for evaluation. /35
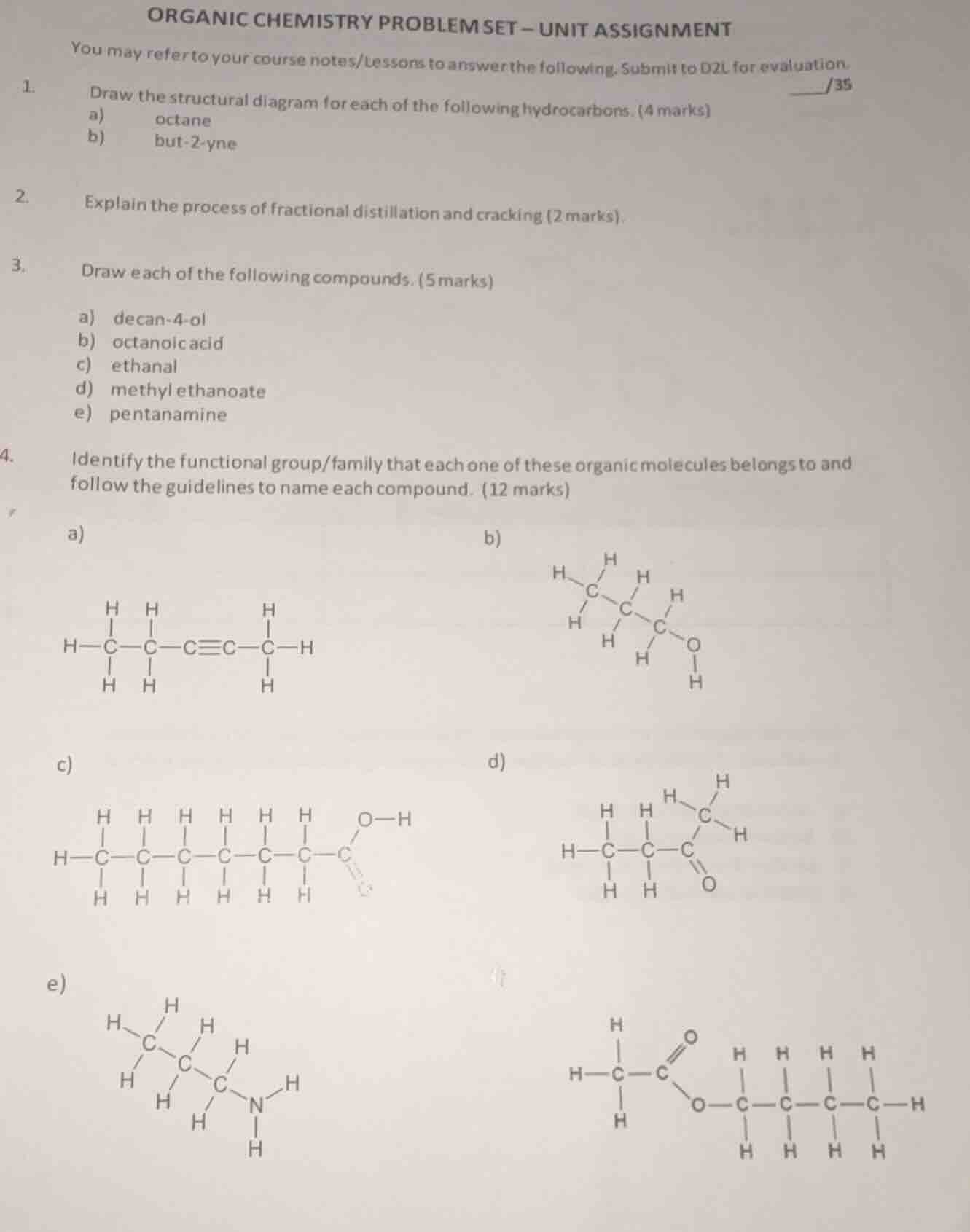
- draw the structural diagram for each of the following hydrocarbons. (4 marks)
a) octane
b) but-2-yne
- explain the process of fractional distillation and cracking (2 marks).
- draw each of the following compounds. (5 marks)
a) decan-4-ol
b) octanoic acid
c) ethanal
d) methyl ethanoate
e) pentanamine
- identify the functional group/family that each one of these organic molecules belongs to and follow the guidelines to name each compound. (12 marks)
a)
b)
c)
d)
e)
1a) Structural Diagram of Octane
Step1: Determine Formula
Octane is an alkane with 8 carbons, formula $C_8H_{18}$.
Step2: Draw Carbon Chain
Draw a straight chain of 8 carbon atoms.
Step3: Add Hydrogen Atoms
Each carbon (except ends) has 2 H, end carbons have 3 H.
Structure:
$$\ce{H_{3}C - CH_{2} - CH_{2} - CH_{2} - CH_{2} - CH_{2} - CH_{2} - CH_{3}}$$
1b) Structural Diagram of But-2-yne
Step1: Determine Formula
But-2-yne is an alkyne with 4 carbons, triple bond at C2, formula $C_4H_6$.
Step2: Draw Carbon Chain
Draw 4 carbons: C1 - C2 ≡ C3 - C4.
Step3: Add Hydrogen Atoms
C1 and C4 have 3 H each? No—alkyne: C1 and C4 (terminal) have 3 H? Wait, but-2-yne: C1: $\ce{CH3}$, C2: $\ce{C≡C}$, C3: $\ce{C}$, C4: $\ce{CH3}$. Wait, no: but-2-yne is $\ce{CH3 - C≡C - CH3}$.
Structure:
$$\ce{CH_{3} - C≡C - CH_{3}}$$
2. Fractional Distillation and Cracking
- Fractional Distillation: Separates crude oil into fractions (e.g., gasoline, diesel) by boiling point. Oil is heated, vapor rises in a fractionating column (cooler at top). Vapors condense at different heights (lower BP = higher up).
- Cracking: Breaks large hydrocarbon molecules (e.g., heavy oil) into smaller ones (e.g., gasoline) by heat (thermal) or catalyst (catalytic). Increases yield of useful fuels.
3a) Decan-4-ol
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Step1: Functional Group
Ester (R-COO-R’).
Step2: Parent Chain
Ethanoate (from ethanoic acid) and butyl (from butanol). So methyl ethanoate? Wait, the structure: $\ce{CH3COOCH2CH2CH2CH3}$ (butyl ethanoate). Functional Group: Ester. Name: Butyl ethanoate.
(Note: Some structures may require more precise counting; the above is a general guide.)