QUESTION IMAGE
Question
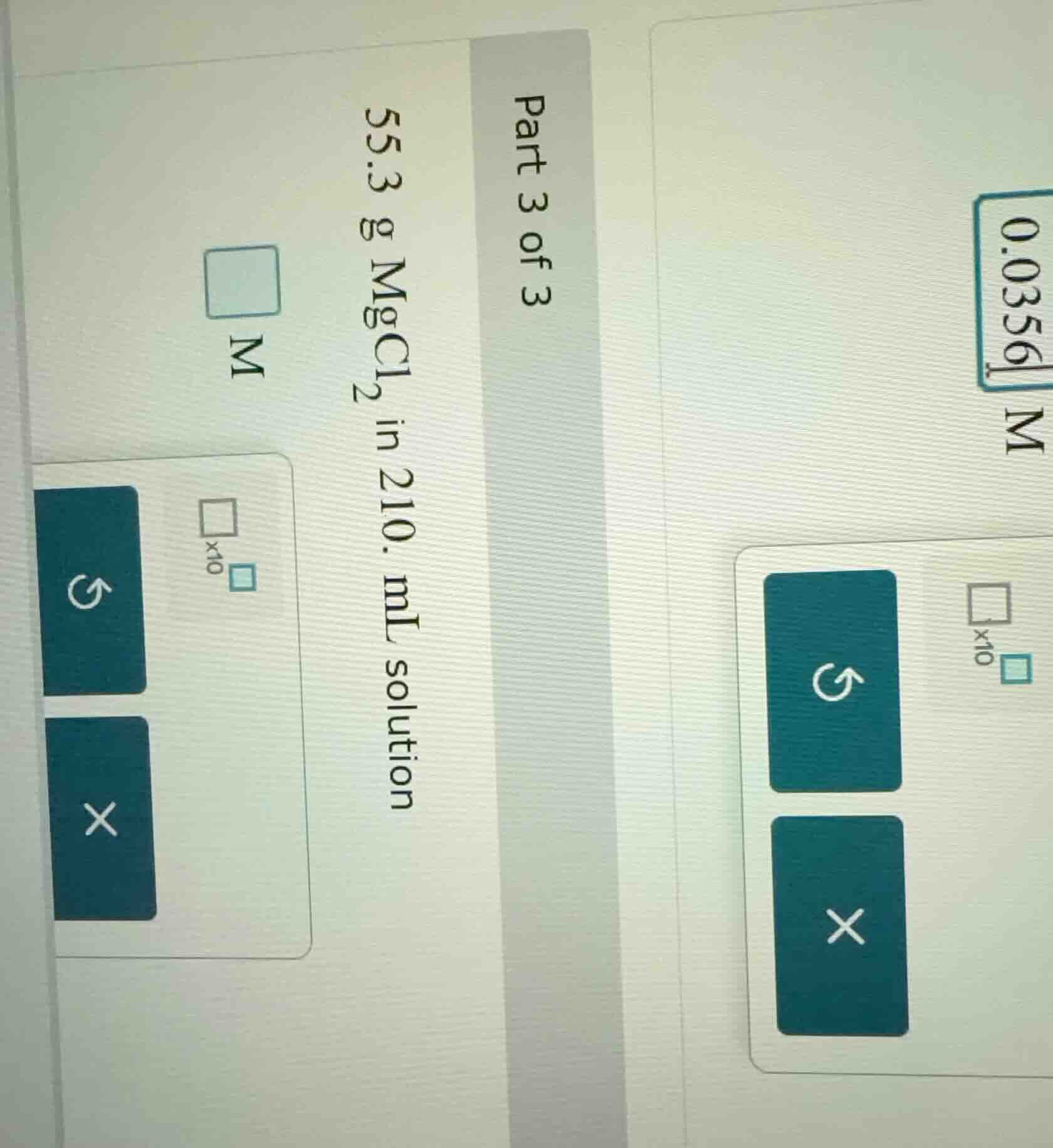
part 3 of 3
55.3 g mgcl₂ in 210. ml solution
□ m
Step1: Calculate molar mass of MgCl₂
Molar mass of Mg = 24.305 g/mol, Cl = 35.453 g/mol.
$$M_{\text{MgCl}_2} = 24.305 + 2\times35.453 = 95.211\ \text{g/mol}$$
Step2: Find moles of MgCl₂
Moles = mass / molar mass.
$$n = \frac{55.3}{95.211} \approx 0.5808\ \text{mol}$$
Step3: Convert volume to liters
$$V = \frac{210}{1000} = 0.210\ \text{L}$$
Step4: Calculate molarity (M)
Molarity = moles / volume.
$$M = \frac{0.5808}{0.210} \approx 2.77$$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
2.77 M