QUESTION IMAGE
Question
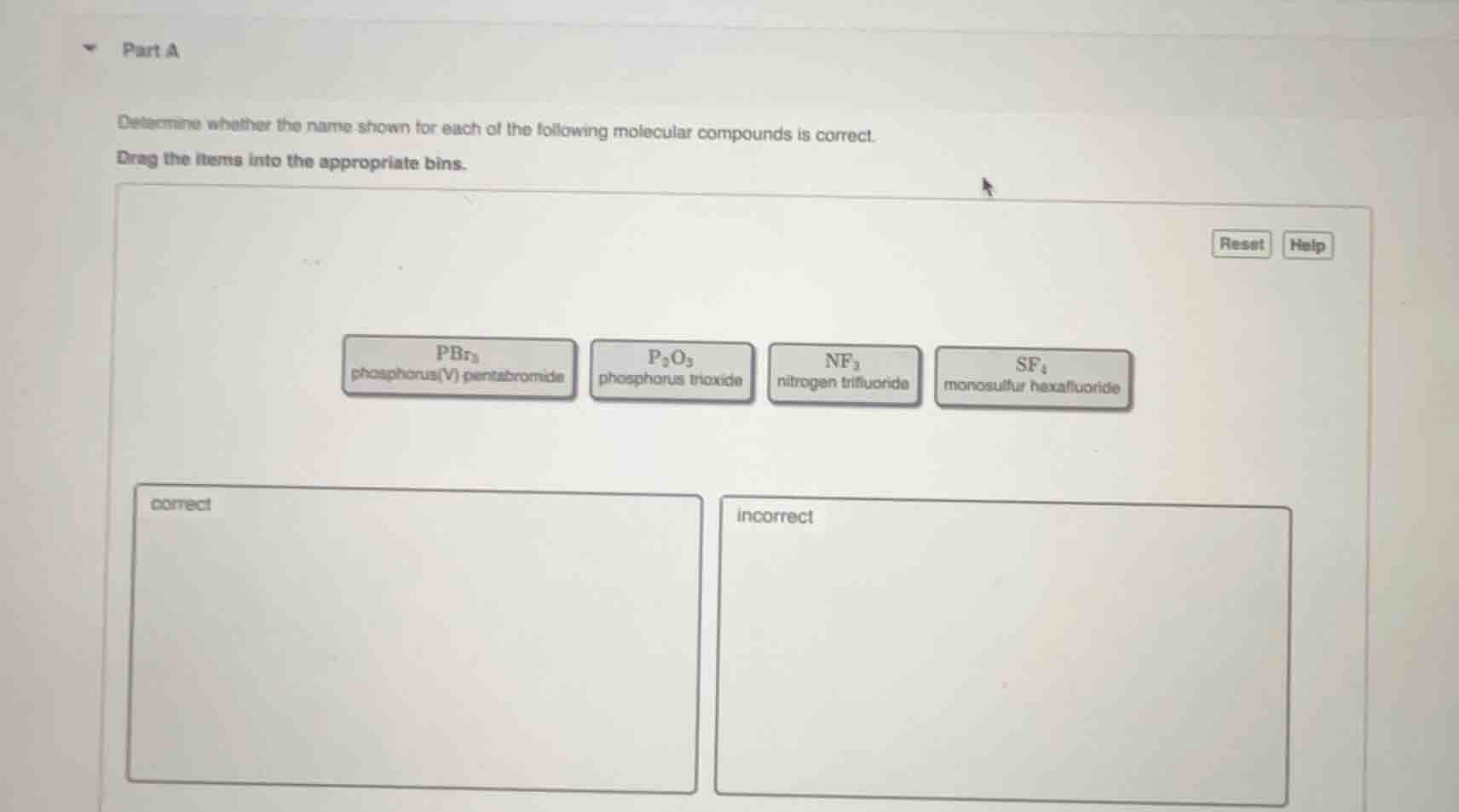
part a
determine whether the name shown for each of the following molecular compounds is correct.
drag the items into the appropriate bins.
pbr₅
phosphorus(v) pentabromide
p₂o₃
phosphorus trioxide
nf₃
nitrogen trifluoride
sf₆
monosulfur hexafluoride
correct
incorrect
Response
To determine the correctness of the names of the molecular compounds, we use the rules for naming molecular compounds (prefixes to indicate the number of atoms, except for the first element if there is only one atom):
1. Analyze \( \ce{PBr_5} \) (phosphorus(V) pentabromide)
- For molecular compounds, we use prefixes (mono-, di-, tri-, etc.) to indicate the number of each atom. However, when the first element has a subscript of 1, we don’t use “mono-”, but for the second element, we do use prefixes.
- \( \ce{PBr_5} \): P is phosphorus, Br is bromine. The name “phosphorus(V) pentabromide” is incorrect for a molecular compound—molecular compounds use prefixes (e.g., “phosphorus pentabromide” without the Roman numeral, since Roman numerals are for ionic compounds with variable oxidation states, but molecular compounds use prefixes to show atom counts). Wait, actually, \( \ce{PBr_5} \) is a molecular compound, and the correct name is “phosphorus pentabromide” (no Roman numeral, as molecular compounds use prefixes, not oxidation states). But let's check the other compounds first.
2. Analyze \( \ce{P_2O_3} \) (phosphorus trioxide)
- For \( \ce{P_2O_3} \): The subscript of P is 2, so we use the prefix “di-” for phosphorus, and subscript of O is 3, so “tri-” for oxygen. The correct name should be “diphosphorus trioxide”, not “phosphorus trioxide” (missing the “di-” prefix for P). Thus, the name is incorrect.
3. Analyze \( \ce{NF_3} \) (nitrogen trifluoride)
- For \( \ce{NF_3} \): N has a subscript of 1 (so no prefix), F has a subscript of 3 (prefix “tri-”). The name “nitrogen trifluoride” is correct (N: 1 atom → no prefix; F: 3 atoms → “tri-”).
4. Analyze \( \ce{SF_6} \) (monosulfur hexafluoride)
- For \( \ce{SF_6} \): S has a subscript of 1. For the first element in a molecular compound, we do not use “mono-” (the prefix “mono-” is only used for the second element if needed). So the correct name is “sulfur hexafluoride” (not “monosulfur hexafluoride”—“mono-” is unnecessary for the first element). Thus, the name is incorrect.
Final Categorization:
- Correct: \( \ce{NF_3} \) (nitrogen trifluoride)
- Incorrect: \( \ce{PBr_5} \) (phosphorus(V) pentabromide), \( \ce{P_2O_3} \) (phosphorus trioxide), \( \ce{SF_6} \) (monosulfur hexafluoride)
Brief Explanations
- \( \ce{NF_3} \): Follows molecular naming rules (N: 1 atom → no prefix; F: 3 atoms → “tri-”).
- \( \ce{PBr_5} \): Uses Roman numeral (for ionic compounds) instead of prefixes (molecular compounds use prefixes).
- \( \ce{P_2O_3} \): Missing “di-” prefix for P (needs “diphosphorus trioxide”).
- \( \ce{SF_6} \): Unnecessary “mono-” prefix for S (first element with subscript 1).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Correct: \( \boldsymbol{\ce{NF_3}} \) (nitrogen trifluoride)
- Incorrect: \( \boldsymbol{\ce{PBr_5}} \) (phosphorus(V) pentabromide), \( \boldsymbol{\ce{P_2O_3}} \) (phosphorus trioxide), \( \boldsymbol{\ce{SF_6}} \) (monosulfur hexafluoride)