QUESTION IMAGE
Question
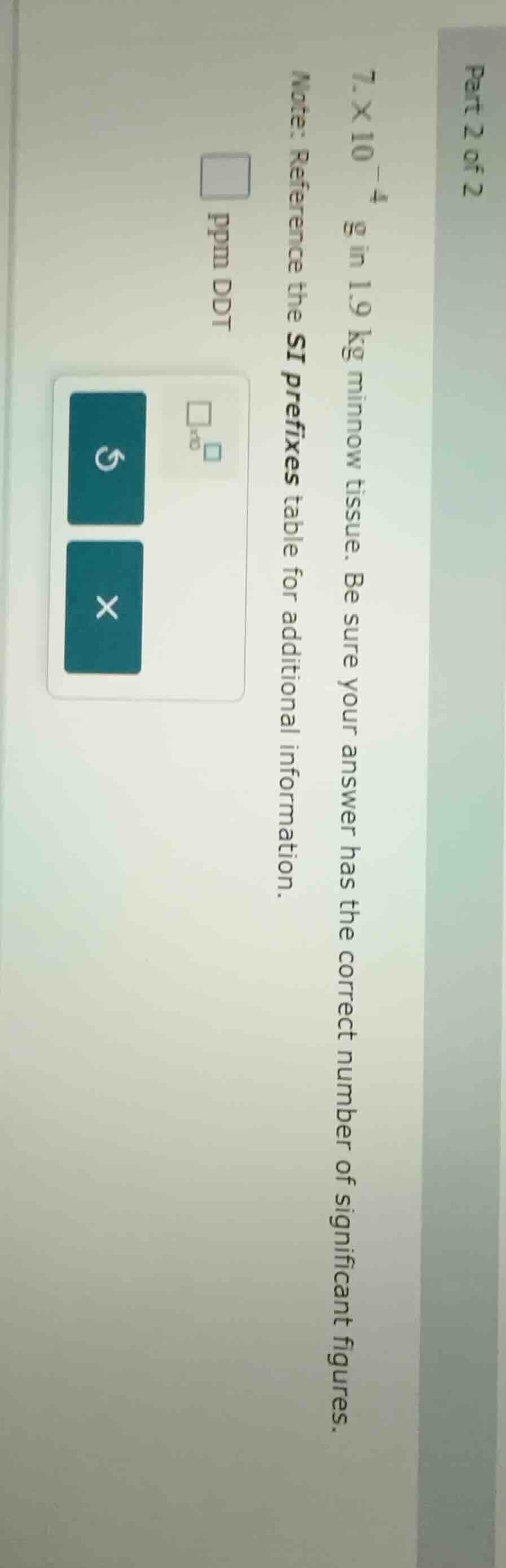
part 2 of 2
$7.\times 10^{-4}$ g in 1.9 kg minnow tissue. be sure your answer has the correct number of significant figures.
note: reference the si prefixes table for additional information.
ppm ddt
Step1: Convert kg to g (SI units)
Since $1\ \text{kg} = 1000\ \text{g}$, $1.9\ \text{kg} = 1.9 \times 10^3\ \text{g}$
Step2: Recall ppm definition
ppm = $\frac{\text{mass of solute (g)}}{\text{mass of solution (g)}} \times 10^6$
Step3: Substitute values into formula
$$\text{ppm DDT} = \frac{7. \times 10^{-4}\ \text{g}}{1.9 \times 10^3\ \text{g}} \times 10^6$$
Step4: Calculate the result
First simplify the expression: $\frac{7. \times 10^{-4} \times 10^6}{1.9 \times 10^3} = \frac{7. \times 10^{2}}{1.9 \times 10^3} = \frac{7.}{1.9 \times 10^{1}} \approx 0.37$
Round to 1 significant figure (from $7. \times 10^{-4}$)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$4 \times 10^{-1}$ ppm DDT (or 0.4 ppm DDT)