QUESTION IMAGE
Question
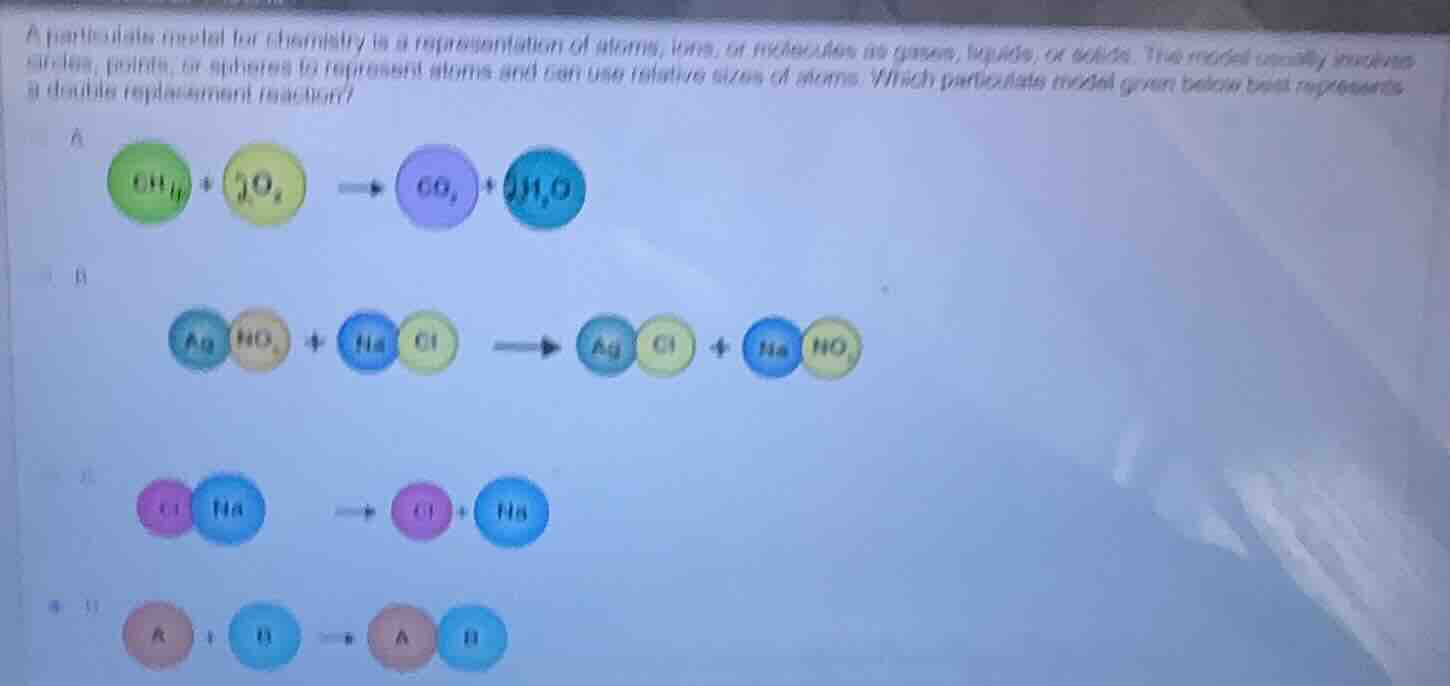
a particulate model for chemistry is a representation of atoms, ions, or molecules in gases, liquids, or solids. the model usually includes circles, points, or spheres to represent atoms and can use relative sizes of atoms. which particulate model given below best represents a double replacement reaction?
a
$\ce{ch_{4} + 2o_{2} -> co_{2} + 2h_{2}o}$
b
$\ce{agno_{3} + nacl -> agcl + nano_{3}}$
c
$\ce{cl_{2} + h_{2} -> 2hcl}$ (note: the original ocr for option c might have a typo, assuming its a reaction like this based on the images atom representation)
d
$\ce{a + b -> ab}$ (assuming the image for d shows a combination reaction)
A double replacement reaction is a chemical reaction where the positive and negative ions of two ionic compounds swap places to form two new compounds.
- Option A shows a combustion reaction (a hydrocarbon reacting with oxygen to form CO₂ and H₂O), which is a redox reaction, not double replacement.
- Option B shows two ionic compounds (AgNO₃ and NaCl) swapping ions: Ag⁺ combines with Cl⁻, and Na⁺ combines with NO₃⁻, forming AgCl and NaNO₃, which fits the definition of a double replacement reaction.
- Option C shows the dissociation of a compound into its ions, not a double replacement.
- Option D shows a combination (synthesis) reaction where two substances join to form one compound, not double replacement.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. $\text{AgNO}_3 + \text{NaCl}
ightarrow \text{AgCl} + \text{NaNO}_3$ (represented by the particulate model of AgNO₃ + NaCl reacting to form AgCl + NaNO₃)