QUESTION IMAGE
Question
phase changes
quiz complete
1 2 3 4 5 6 7 8 9 10
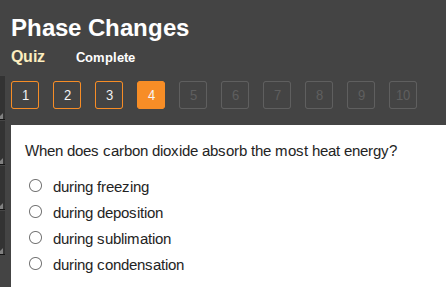
when does carbon dioxide absorb the most heat energy?
○ during freezing
○ during deposition
○ during sublimation
○ during condensation
Sublimation is the phase change of a substance directly from a solid state to a gaseous state. This process requires a large input of heat energy to break the strong intermolecular forces holding the solid structure together, far more than the exothermic (heat-releasing) processes of freezing, deposition, and condensation, which all release heat rather than absorb it.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
during sublimation