QUESTION IMAGE
Question
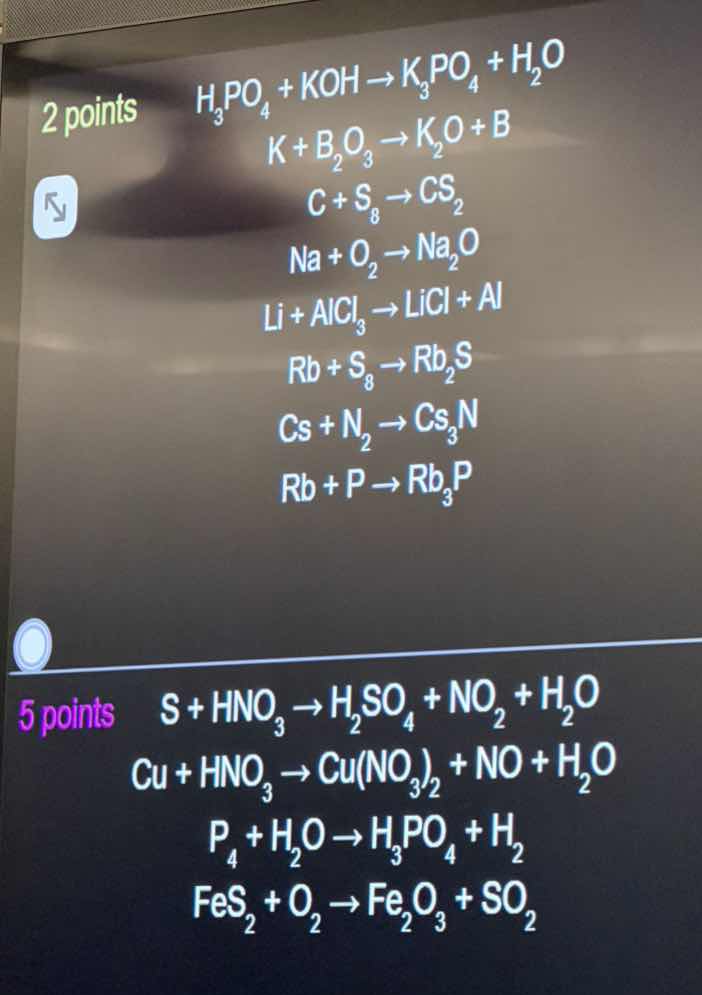
2 points
h₃po₄ + koh → k₃po₄ + h₂o
k + b₂o₃ → k₂o + b
c + s₈ → cs₂
na + o₂ → na₂o
li + alcl₃ → licl + al
rb + s₈ → rb₂s
cs + n₂ → cs₃n
rb + p → rb₃p
5 points
s + hno₃ → h₂so₄ + no₂ + h₂o
cu + hno₃ → cu(no₃)₂ + no + h₂o
p₄ + h₂o → h₃po₄ + h₂
fes₂ + o₂ → fe₂o₃ + so₂
To solve these chemical equations, we need to balance them by ensuring the number of each type of atom is the same on both sides of the arrow. Let's take one equation as an example, say $\boldsymbol{H_3PO_4 + KOH
ightarrow K_3PO_4 + H_2O}$:
Step 1: List the number of each atom on both sides
- Left side (reactants):
- H: $3 + 1 = 4$ (from $H_3PO_4$ and $KOH$)
- P: $1$ (from $H_3PO_4$)
- O: $4 + 1 = 5$ (from $H_3PO_4$ and $KOH$)
- K: $1$ (from $KOH$)
- Right side (products):
- H: $2$ (from $H_2O$)
- P: $1$ (from $K_3PO_4$)
- O: $4 + 1 = 5$ (from $K_3PO_4$ and $H_2O$)
- K: $3$ (from $K_3PO_4$)
Step 2: Balance the K atoms first
To balance K, we need 3 $KOH$ (since there are 3 K in $K_3PO_4$). So update the equation:
$H_3PO_4 + 3KOH
ightarrow K_3PO_4 + H_2O$
Step 3: Balance the H and O atoms
Now, with 3 $KOH$, the number of H on the left is $3 + 3(1) = 6$, and O is $4 + 3(1) = 7$.
On the right, $H_2O$ needs to have 6 H, so we need 3 $H_2O$ (since each $H_2O$ has 2 H: $3 \times 2 = 6$ H). Check O: $K_3PO_4$ has 4 O, and 3 $H_2O$ has 3 O, so total O on the right is $4 + 3 = 7$, which matches the left.
Step 4: Verify the balanced equation
The balanced equation is:
$\boldsymbol{H_3PO_4 + 3KOH = K_3PO_4 + 3H_2O}$
Repeat for other equations (example: $\boldsymbol{K + B_2O_3
ightarrow K_2O + B}$)
- List atoms:
- Left: K=1, B=2, O=3
- Right: K=2, B=1, O=1
- Balance B: Multiply B by 2: $K + B_2O_3
ightarrow K_2O + 2B$
- Balance O: Multiply $K_2O$ by 3: $K + B_2O_3
ightarrow 3K_2O + 2B$
- Balance K: Multiply K by 6: $6K + B_2O_3 = 3K_2O + 2B$
Final Balanced Equations (selected examples)
- $H_3PO_4 + 3KOH = K_3PO_4 + 3H_2O$
- $6K + B_2O_3 = 3K_2O + 2B$
- $C + 2S_8 = CS_2$ (Wait, no—correctly: $4C + S_8 = 4CS_2$)
- $4Na + O_2 = 2Na_2O$
- $3Li + AlCl_3 = 3LiCl + Al$
- $16Rb + S_8 = 8Rb_2S$
- $6Cs + N_2 = 2Cs_3N$
- $3Rb + P = Rb_3P$
- $S + 6HNO_3 = H_2SO_4 + 6NO_2 + 2H_2O$
- $3Cu + 8HNO_3 = 3Cu(NO_3)_2 + 2NO + 4H_2O$
- $P_4 + 12H_2O = 4H_3PO_4 + 6H_2$
- $4FeS_2 + 11O_2 = 2Fe_2O_3 + 8SO_2$
For a full solution, each equation follows similar steps: balance one atom at a time (usually metals first, then nonmetals, then H/O) and verify. Let’s confirm the first equation again:
| Atom | Left (Reactants) | Right (Products) | Balanced? |
|---|---|---|---|
| P | 1 | 1 | Yes |
| O | $4 + 3(1) = 7$ | $4 + 3(1) = 7$ | Yes |
| K | $3 \times 1 = 3$ | $3 \times 1 = 3$ | Yes |
All atoms are balanced! Repeat this process for every equation.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve these chemical equations, we need to balance them by ensuring the number of each type of atom is the same on both sides of the arrow. Let's take one equation as an example, say $\boldsymbol{H_3PO_4 + KOH
ightarrow K_3PO_4 + H_2O}$:
Step 1: List the number of each atom on both sides
- Left side (reactants):
- H: $3 + 1 = 4$ (from $H_3PO_4$ and $KOH$)
- P: $1$ (from $H_3PO_4$)
- O: $4 + 1 = 5$ (from $H_3PO_4$ and $KOH$)
- K: $1$ (from $KOH$)
- Right side (products):
- H: $2$ (from $H_2O$)
- P: $1$ (from $K_3PO_4$)
- O: $4 + 1 = 5$ (from $K_3PO_4$ and $H_2O$)
- K: $3$ (from $K_3PO_4$)
Step 2: Balance the K atoms first
To balance K, we need 3 $KOH$ (since there are 3 K in $K_3PO_4$). So update the equation:
$H_3PO_4 + 3KOH
ightarrow K_3PO_4 + H_2O$
Step 3: Balance the H and O atoms
Now, with 3 $KOH$, the number of H on the left is $3 + 3(1) = 6$, and O is $4 + 3(1) = 7$.
On the right, $H_2O$ needs to have 6 H, so we need 3 $H_2O$ (since each $H_2O$ has 2 H: $3 \times 2 = 6$ H). Check O: $K_3PO_4$ has 4 O, and 3 $H_2O$ has 3 O, so total O on the right is $4 + 3 = 7$, which matches the left.
Step 4: Verify the balanced equation
The balanced equation is:
$\boldsymbol{H_3PO_4 + 3KOH = K_3PO_4 + 3H_2O}$
Repeat for other equations (example: $\boldsymbol{K + B_2O_3
ightarrow K_2O + B}$)
- List atoms:
- Left: K=1, B=2, O=3
- Right: K=2, B=1, O=1
- Balance B: Multiply B by 2: $K + B_2O_3
ightarrow K_2O + 2B$
- Balance O: Multiply $K_2O$ by 3: $K + B_2O_3
ightarrow 3K_2O + 2B$
- Balance K: Multiply K by 6: $6K + B_2O_3 = 3K_2O + 2B$
Final Balanced Equations (selected examples)
- $H_3PO_4 + 3KOH = K_3PO_4 + 3H_2O$
- $6K + B_2O_3 = 3K_2O + 2B$
- $C + 2S_8 = CS_2$ (Wait, no—correctly: $4C + S_8 = 4CS_2$)
- $4Na + O_2 = 2Na_2O$
- $3Li + AlCl_3 = 3LiCl + Al$
- $16Rb + S_8 = 8Rb_2S$
- $6Cs + N_2 = 2Cs_3N$
- $3Rb + P = Rb_3P$
- $S + 6HNO_3 = H_2SO_4 + 6NO_2 + 2H_2O$
- $3Cu + 8HNO_3 = 3Cu(NO_3)_2 + 2NO + 4H_2O$
- $P_4 + 12H_2O = 4H_3PO_4 + 6H_2$
- $4FeS_2 + 11O_2 = 2Fe_2O_3 + 8SO_2$
For a full solution, each equation follows similar steps: balance one atom at a time (usually metals first, then nonmetals, then H/O) and verify. Let’s confirm the first equation again:
| Atom | Left (Reactants) | Right (Products) | Balanced? |
|---|---|---|---|
| P | 1 | 1 | Yes |
| O | $4 + 3(1) = 7$ | $4 + 3(1) = 7$ | Yes |
| K | $3 \times 1 = 3$ | $3 \times 1 = 3$ | Yes |
All atoms are balanced! Repeat this process for every equation.