QUESTION IMAGE
Question
possible points: 4
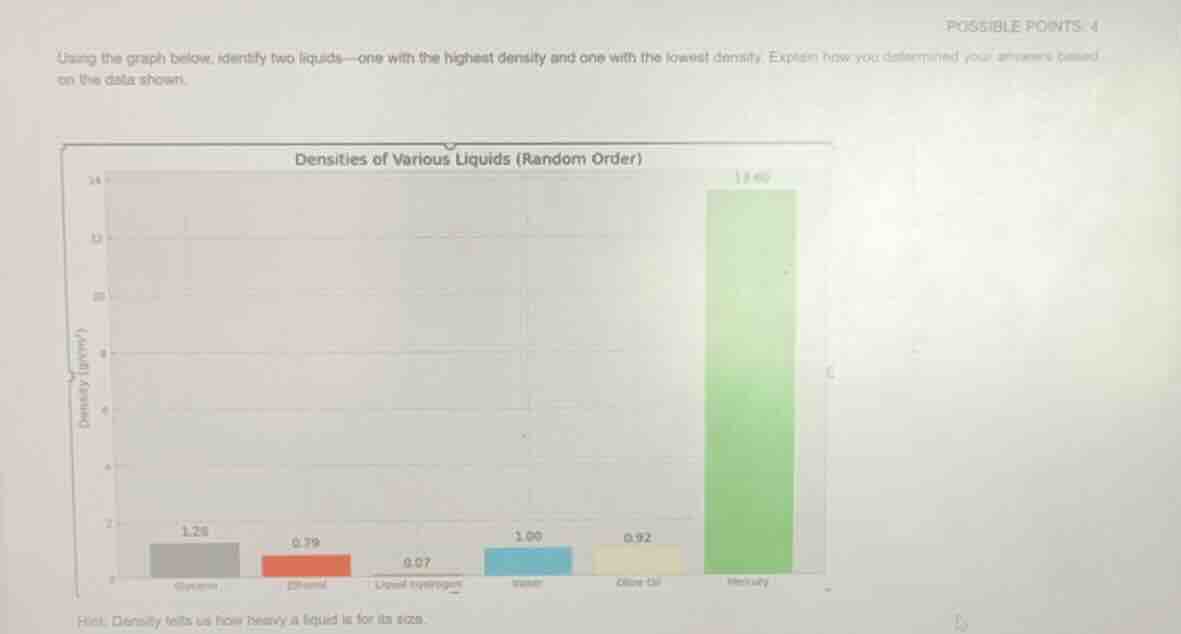
using the graph below, identify two liquids—one with the highest density and one with the lowest density. explain how you determined your answers based on the data shown.
densities of various liquids (random order)
hint: density tells us how heavy a liquid is for its size.
Brief Explanations
- To find the liquid with the highest density, locate the tallest bar on the bar graph, as it corresponds to the highest density value. The tallest bar is labeled Mercury, with a density of 13.60 g/mL.
- To find the liquid with the lowest density, locate the shortest bar on the bar graph, as it corresponds to the lowest density value. The shortest bar is labeled Liquid Hydrogen, with a density of 0.07 g/mL.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Highest density liquid: Mercury (density = 13.60 g/mL), determined by its being the tallest bar on the graph.
- Lowest density liquid: Liquid Hydrogen (density = 0.07 g/mL), determined by its being the shortest bar on the graph.