QUESTION IMAGE
Question
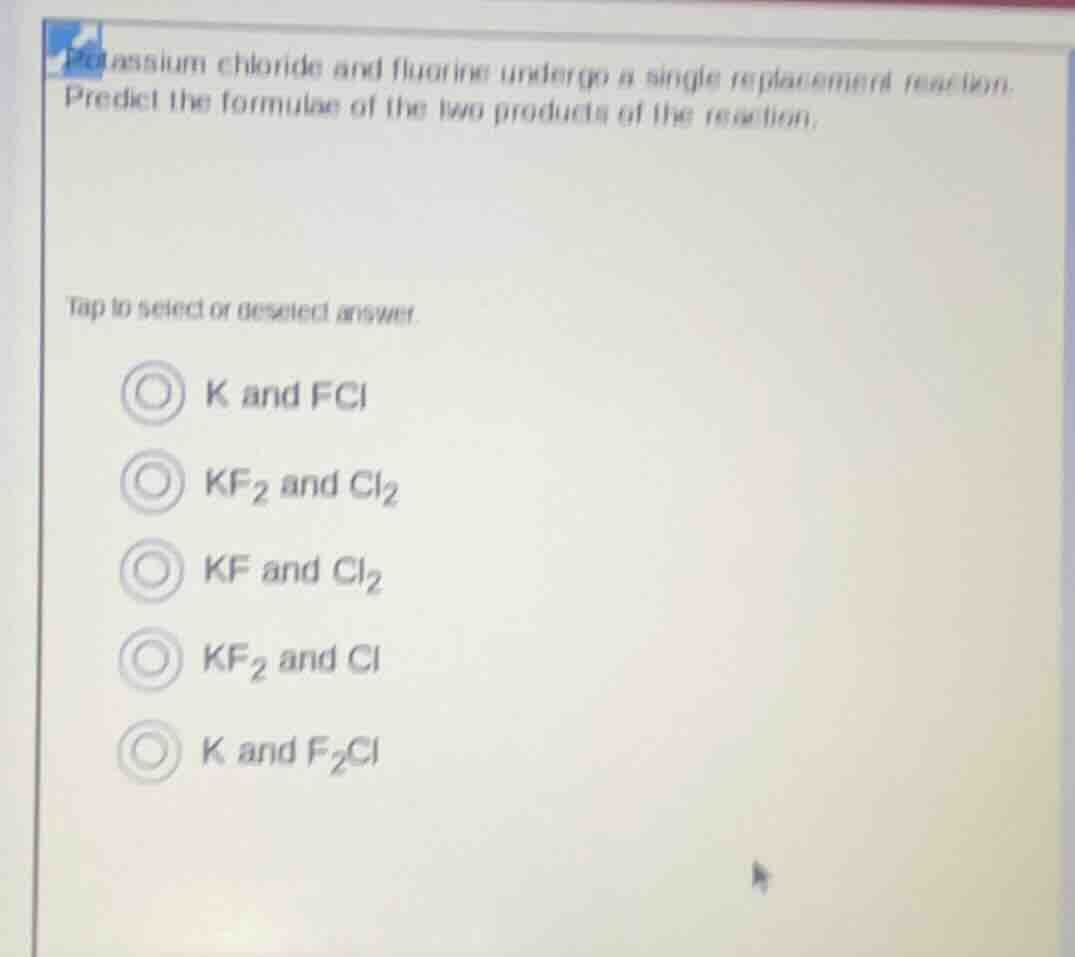
potassium chloride and fluorine undergo a single replacement reaction. predict the formulae of the two products of the reaction. tap to select or deselect answer. k and fcl kf₂ and cl₂ kf and cl₂ kf₂ and cl k and f₂cl
Step1: Recall Single Replacement Reaction
In a single replacement reaction, a more reactive element replaces a less reactive element in a compound. The activity series for halogens (Group 17) is \( \text{F}_2 > \text{Cl}_2 > \text{Br}_2 > \text{I}_2 \), so fluorine (\( \text{F}_2 \)) is more reactive than chlorine (\( \text{Cl}_2 \)). The reactants are potassium chloride (\( \text{KCl} \)) and fluorine (\( \text{F}_2 \)).
Step2: Write the Reaction
The general form for single replacement (halogen replacing halogen) is \( \text{X}_2 + 2\text{Y}\text{Z}
ightarrow 2\text{Y}\text{X} + \text{Z}_2 \) (where \( \text{X} \) is more reactive than \( \text{Z} \)). Here, \( \text{X} = \text{F} \), \( \text{Y} = \text{K} \), \( \text{Z} = \text{Cl} \). So the reaction is \( \text{F}_2 + 2\text{KCl}
ightarrow 2\text{KF} + \text{Cl}_2 \).
Step3: Analyze Products
From the reaction, the products are potassium fluoride (\( \text{KF} \)) and chlorine gas (\( \text{Cl}_2 \)). Checking the options, "KF and \( \text{Cl}_2 \)" matches.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. KF and \( \text{Cl}_2 \) (assuming the third option is labeled C, with the text "KF and \( \text{Cl}_2 \)")