QUESTION IMAGE
Question
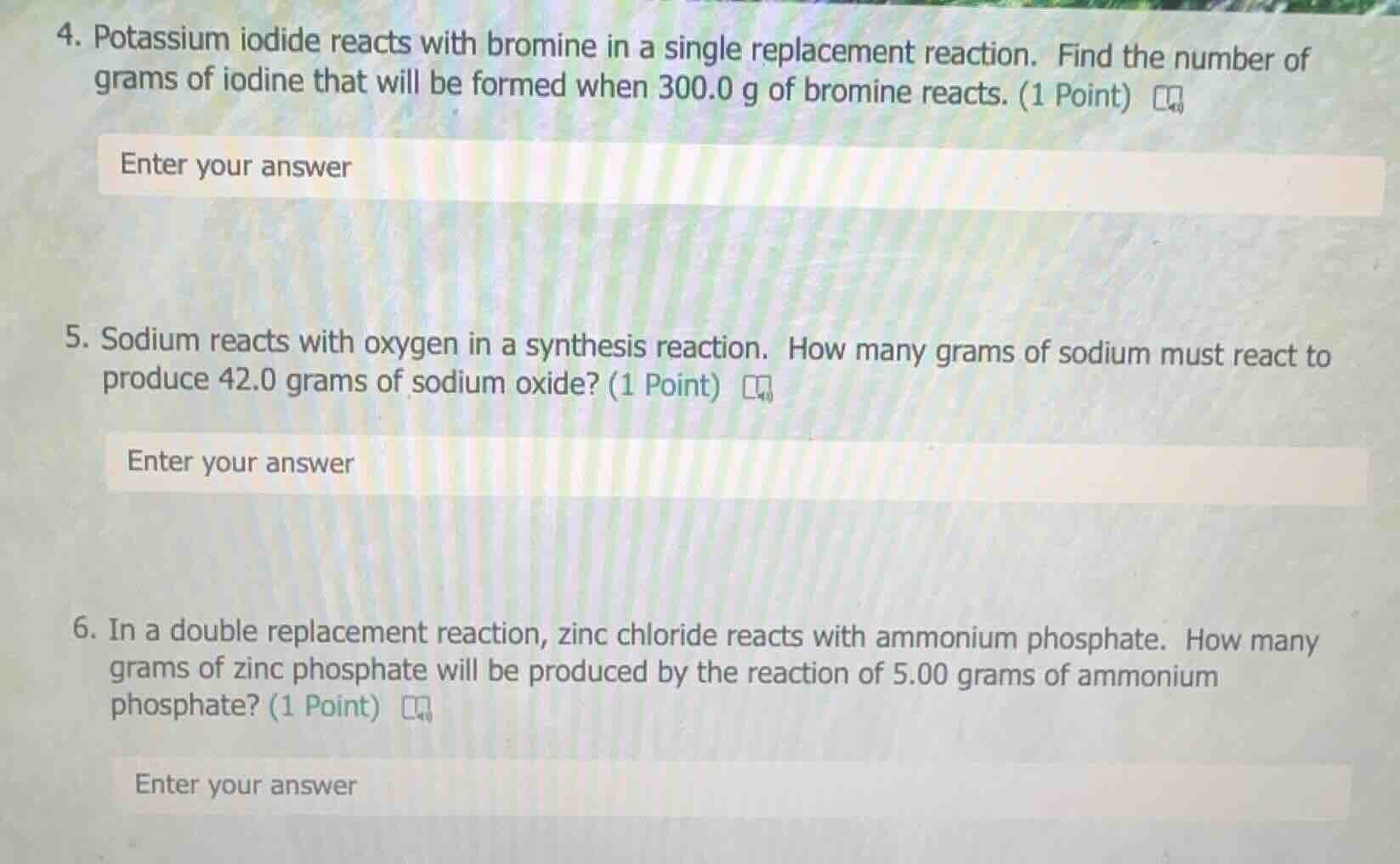
- potassium iodide reacts with bromine in a single replacement reaction. find the number of grams of iodine that will be formed when 300.0 g of bromine reacts. (1 point) enter your answer 5. sodium reacts with oxygen in a synthesis reaction. how many grams of sodium must react to produce 42.0 grams of sodium oxide? (1 point) enter your answer 6. in a double replacement reaction, zinc chloride reacts with ammonium phosphate. how many grams of zinc phosphate will be produced by the reaction of 5.00 grams of ammonium phosphate? (1 point) enter your answer
Question 4
Step1: Write the balanced reaction
The reaction between potassium iodide ($\ce{KI}$) and bromine ($\ce{Br_2}$) is a single - replacement reaction. The balanced chemical equation is:
Step2: Calculate moles of $\ce{Br_2}$
The molar mass of $\ce{Br_2}$ is $M(\ce{Br_2}) = 2\times79.90\space g/mol=159.8\space g/mol$.
Given mass of $\ce{Br_2}$, $m = 300.0\space g$.
Moles of $\ce{Br_2}$, $n=\frac{m}{M}=\frac{300.0\space g}{159.8\space g/mol}\approx1.877\space mol$
Step3: Determine moles of $\ce{I_2}$ formed
From the balanced equation, the mole ratio of $\ce{Br_2}$ to $\ce{I_2}$ is $1:1$. So moles of $\ce{I_2}$ formed, $n(\ce{I_2})=n(\ce{Br_2}) = 1.877\space mol$
Step4: Calculate mass of $\ce{I_2}$
The molar mass of $\ce{I_2}$ is $M(\ce{I_2})=2\times126.90\space g/mol = 253.8\space g/mol$
Mass of $\ce{I_2}$, $m=n\times M=1.877\space mol\times253.8\space g/mol\approx476.4\space g$
Step1: Write the balanced reaction
The synthesis reaction between sodium ($\ce{Na}$) and oxygen ($\ce{O_2}$) to form sodium oxide ($\ce{Na_2O}$) has the balanced equation:
Step2: Calculate moles of $\ce{Na_2O}$
The molar mass of $\ce{Na_2O}$ is $M(\ce{Na_2O})=2\times22.99 + 16.00=61.98\space g/mol$
Given mass of $\ce{Na_2O}$, $m = 42.0\space g$
Moles of $\ce{Na_2O}$, $n=\frac{m}{M}=\frac{42.0\space g}{61.98\space g/mol}\approx0.6776\space mol$
Step3: Determine moles of $\ce{Na}$
From the balanced equation, the mole ratio of $\ce{Na}$ to $\ce{Na_2O}$ is $4:2 = 2:1$. So moles of $\ce{Na}$, $n(\ce{Na}) = 2\times n(\ce{Na_2O})$
$n(\ce{Na})=2\times0.6776\space mol = 1.3552\space mol$
Step4: Calculate mass of $\ce{Na}$
The molar mass of $\ce{Na}$ is $M(\ce{Na}) = 22.99\space g/mol$
Mass of $\ce{Na}$, $m=n\times M=1.3552\space mol\times22.99\space g/mol\approx31.16\space g$
Step1: Write the balanced reaction
The double - replacement reaction between zinc chloride ($\ce{ZnCl_2}$) and ammonium phosphate ($\ce{(NH_4)_3PO_4}$) has the balanced equation:
Step2: Calculate moles of $\ce{(NH_4)_3PO_4}$
The molar mass of $\ce{(NH_4)_3PO_4}$ is $M = 3\times(14.01 + 4\times1.008)+30.97+4\times16.00$
$M=3\times(14.01 + 4.032)+30.97 + 64.00$
$M=3\times18.042+30.97 + 64.00$
$M = 54.126+30.97+64.00=149.096\space g/mol$
Given mass of $\ce{(NH_4)_3PO_4}$, $m = 5.00\space g$
Moles of $\ce{(NH_4)_3PO_4}$, $n=\frac{m}{M}=\frac{5.00\space g}{149.096\space g/mol}\approx0.03354\space mol$
Step3: Determine moles of $\ce{Zn_3(PO_4)_2}$ formed
From the balanced equation, the mole ratio of $\ce{(NH_4)_3PO_4}$ to $\ce{Zn_3(PO_4)_2}$ is $2:1$. So moles of $\ce{Zn_3(PO_4)_2}$, $n(\ce{Zn_3(PO_4)_2})=\frac{1}{2}\times n(\ce{(NH_4)_3PO_4})$
$n(\ce{Zn_3(PO_4)_2})=\frac{1}{2}\times0.03354\space mol = 0.01677\space mol$
Step4: Calculate mass of $\ce{Zn_3(PO_4)_2}$
The molar mass of $\ce{Zn_3(PO_4)_2}$ is $M=3\times65.38+2\times(30.97 + 4\times16.00)$
$M = 196.14+2\times(30.97 + 64.00)$
$M=196.14 + 2\times94.97$
$M=196.14+189.94 = 386.08\space g/mol$
Mass of $\ce{Zn_3(PO_4)_2}$, $m=n\times M=0.01677\space mol\times386.08\space g/mol\approx6.47\space g$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$476.4\space g$