QUESTION IMAGE
Question
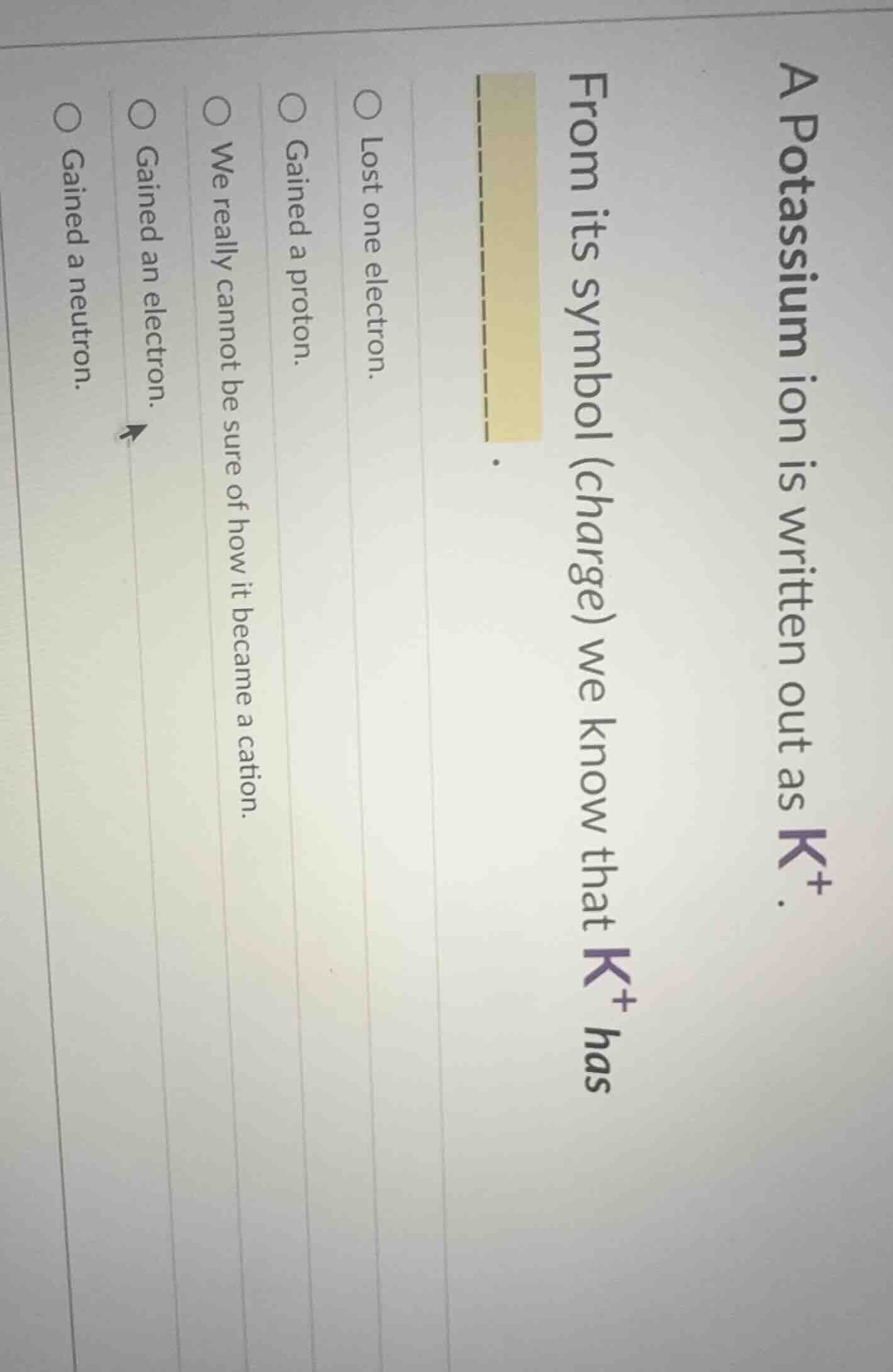
a potassium ion is written out as $k^+$.
from its symbol (charge) we know that $k^+$ has
$\boldsymbol{__________}$.
○ lost one electron.
○ gained a proton.
○ we really cannot be sure of how it became a cation.
○ gained an electron.
○ gained a neutron.
A neutral atom has equal protons and electrons. A positive charge ($+$) on an ion means the atom has more protons than electrons, which occurs when it loses an electron. Protons determine the element, so potassium cannot gain a proton (it would become a different element). Neutron changes affect mass, not charge.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Lost one electron.