QUESTION IMAGE
Question
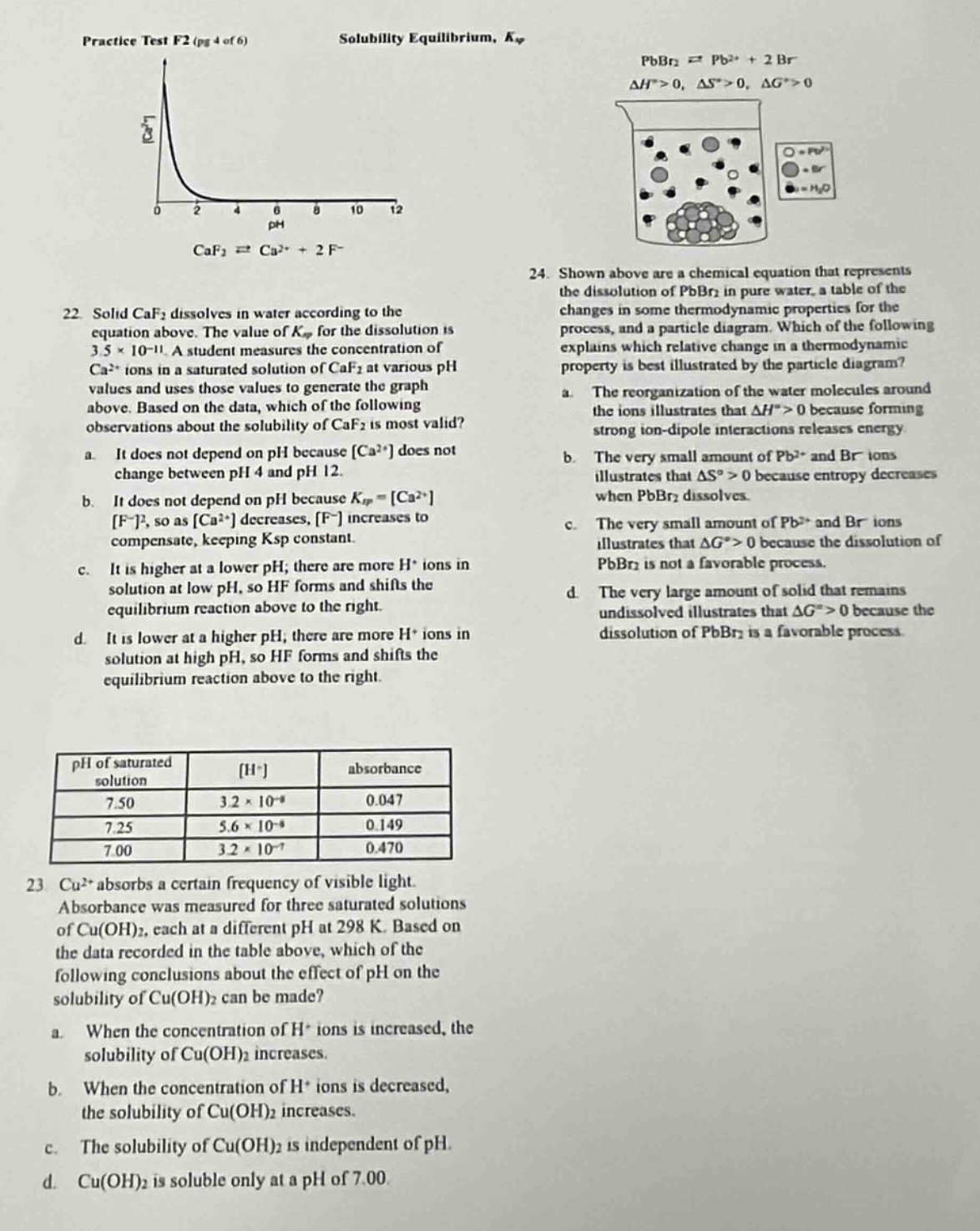
practice test f2 (pg 4 of 6) solubility equilibrium, ( k_{sp} )
( ce{caf_{2}}
ightleftharpoons ce{ca^{2+}} + 2ce{f^{-}} )
- solid ( ce{caf_{2}} ) dissolves in water according to the equation above. the value of ( k_{sp} ) for the dissolution is ( 3.5 \times 10^{-11} ). a student measures the concentration of ( ce{ca^{2+}} ) ions in a saturated solution of ( ce{caf_{2}} ) at various ph values and uses those values to generate the graph above. based on the data, which of the following observations about the solubility of ( ce{caf_{2}} ) is most valid?
a. it does not depend on ph because ( ce{ca^{2+}} ) does not change between ph 4 and ph 12.
b. it does not depend on ph because ( k_{sp} = ce{ca^{2+}}ce{f^{-}}^2 ), so as ( ce{ca^{2+}} ) decreases, ( ce{f^{-}} ) increases to compensate, keeping ( k_{sp} ) constant.
c. it is higher at a lower ph; there are more ( ce{h^{+}} ) ions in solution at low ph, so hf forms and shifts the equilibrium reaction above to the right.
d. it is lower at a higher ph; there are more ( ce{h^{+}} ) ions in solution at high ph, so hf forms and shifts the equilibrium reaction above to the right.
| ph of saturated solution | ( ce{h^{+}} ) | absorbance |
|---|---|---|
| 7.25 | ( 5.6 \times 10^{-8} ) | 0.149 |
| 7.00 | ( 3.2 \times 10^{-7} ) | 0.470 |
- ( ce{cu^{2+}} ) absorbs a certain frequency of visible light. absorbance was measured for three saturated solutions of ( ce{cu(oh)_{2}} ), each at a different ph at 298 k. based on the data recorded in the table above, which of the following conclusions about the effect of ph on the solubility of ( ce{cu(oh)_{2}} ) can be made?
a. when the concentration of ( ce{h^{+}} ) ions is increased, the solubility of ( ce{cu(oh)_{2}} ) increases.
b. when the concentration of ( ce{h^{+}} ) ions is decreased, the solubility of ( ce{cu(oh)_{2}} ) increases.
c. the solubility of ( ce{cu(oh)_{2}} ) is independent of ph.
d. ( ce{cu(oh)_{2}} ) is soluble only at a ph of 7.00.
( ce{pbbr_{2}}
ightleftharpoons ce{pb^{2+}} + 2ce{br^{-}} )
( delta h^{circ} > 0 ), ( delta s^{circ} > 0 ), ( delta g^{circ} > 0 )
- shown above are a chemical equation that represents the dissolution of ( ce{pbbr_{2}} ) in pure water, a table of the changes in some thermodynamic properties for the process, and a particle diagram. which of the following explains which relative change in a thermodynamic property is best illustrated by the particle diagram?
a. the reorganization of the water molecules around the ions illustrates that ( delta h^{circ} > 0 ) because forming strong ion - dipole interactions releases energy.
b. the very small amount of ( ce{pb^{2+}} ) and ( ce{br^{-}} ) ions illustrates that ( delta s^{circ} > 0 ) because entropy decreases when ( ce{pbbr_{2}} ) dissolves.
c. the very small amount of ( ce{pb^{2+}} ) and ( ce{br^{-}} ) ions illustrates that ( delta g^{circ} > 0 ) because the dissolution of ( ce{pbbr_{2}} ) is not a favorable process.
d. the very large amount of solid that remains undissolved illustrates that ( delta g^{circ} > 0 ) because the dissolution of ( ce{pbbr_{2}} ) is a favorable process.
Question 22
- Option a: From the graph, \([Ca^{2+}]\) (related to solubility) changes with pH, so solubility depends on pH. Eliminate a.
- Option b: \(K_{sp} = [Ca^{2+}][F^-]^2\), but pH affects \([F^-]\) (via \(F^- + H^+
ightleftharpoons HF\)), so \([Ca^{2+}]\) changes with pH, meaning solubility depends on pH. Eliminate b.
- Option c: At lower pH, \([H^+]\) is higher. \(F^- + H^+
ightleftharpoons HF\) (HF is a weak acid), so \([F^-]\) decreases. The equilibrium \(CaF_2
ightleftharpoons Ca^{2+} + 2F^-\) shifts right (Le Chatelier’s principle), increasing solubility. This matches.
- Option d: Higher pH means lower \([H^+]\), so \([F^-]\) is higher, shifting equilibrium left (decreasing solubility), not right. Eliminate d.
- The dissolution of \(Cu(OH)_2\) is \(Cu(OH)_2
ightleftharpoons Cu^{2+} + 2OH^-\). \(H^+ + OH^-
ightleftharpoons H_2O\), so \([H^+]\) (pH) affects \([OH^-]\).
- From the table: as \([H^+]\) increases (pH decreases, e.g., from 7.50 to 7.00), absorbance (related to \([Cu^{2+}]\), hence solubility) increases.
- Option a: Increasing \([H^+]\) (lower pH) increases solubility (higher absorbance), so this is correct.
- Option b: Decreasing \([H^+]\) (higher pH) would decrease solubility (opposite of data), eliminate b.
- Option c: Data shows solubility changes with pH, eliminate c.
- Option d: The table shows solubility at multiple pHs, not just 7.00, eliminate d.
- Given \(\Delta G^\circ > 0\) (non - spontaneous dissolution, since \(\Delta G^\circ > 0\) for non - favorable processes at standard conditions).
- Option a: \(\Delta H^\circ > 0\) means the process is endothermic. Forming ion - dipole interactions (exothermic) would mean \(\Delta H^\circ < 0\) if that were the main factor, but \(\Delta H^\circ > 0\) here. The particle diagram doesn't show water reorganization related to \(\Delta H^\circ\) in this way. Eliminate a.
- Option b: Dissolution of a solid into ions increases entropy (\(\Delta S^\circ > 0\) should mean more disorder). But the diagram shows few ions, but entropy change for dissolution: solid to ions is an increase in entropy. The reasoning here says entropy decreases, which is wrong. Eliminate b.
- Option c: \(\Delta G^\circ > 0\) means the dissolution is non - favorable (non - spontaneous at standard conditions). The particle diagram shows a large amount of solid (little dissolution), so few \(Pb^{2+}\) and \(Br^-\) ions, indicating dissolution is not favorable, matching \(\Delta G^\circ > 0\).
- Option d: \(\Delta G^\circ > 0\) means dissolution is non - favorable, but this option says it's favorable. Contradiction. Eliminate d.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
c. It is higher at a lower pH; there are more \(H^+\) ions in solution at low pH, so HF forms and shifts the equilibrium reaction above to the right.