QUESTION IMAGE
Question
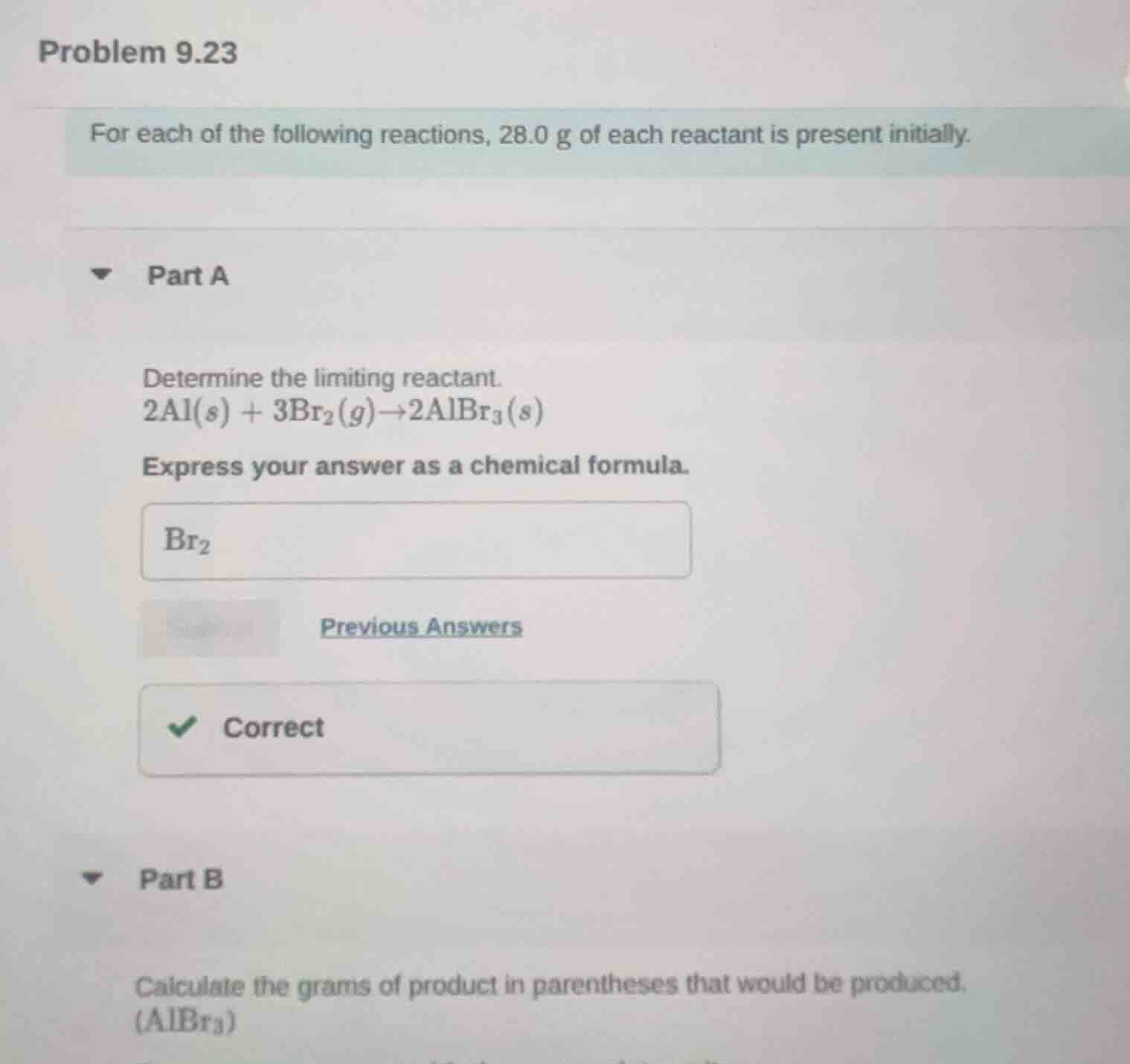
problem 9.23
for each of the following reactions, 28.0 g of each reactant is present initially.
part a
determine the limiting reactant.
$2\text{al}(s) + 3\text{br}_2(g)\
ightarrow2\text{albr}_3(s)$
express your answer as a chemical formula.
$\text{br}_2$
previous answers
correct
part b
calculate the grams of product in parentheses that would be produced.
$(\text{albr}_3)$
Step1: Calculate moles of $\text{Br}_2$
Molar mass of $\text{Br}_2 = 2 \times 79.90 = 159.80\ \text{g/mol}$
$\text{Moles of Br}_2 = \frac{28.0\ \text{g}}{159.80\ \text{g/mol}} \approx 0.1752\ \text{mol}$
Step2: Mole ratio to product
From reaction: $3\ \text{mol Br}_2
ightarrow 2\ \text{mol AlBr}_3$
$\text{Moles of AlBr}_3 = 0.1752\ \text{mol Br}_2 \times \frac{2\ \text{mol AlBr}_3}{3\ \text{mol Br}_2} \approx 0.1168\ \text{mol}$
Step3: Molar mass of $\text{AlBr}_3$
$\text{Molar mass} = 26.98 + 3 \times 79.90 = 26.98 + 239.70 = 266.68\ \text{g/mol}$
Step4: Mass of $\text{AlBr}_3$
$\text{Mass} = 0.1168\ \text{mol} \times 266.68\ \text{g/mol} \approx 31.1\ \text{g}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$31.1\ \text{g}$