QUESTION IMAGE
Question
proton
neutron
electron
nucleus
the number of protons in an atom is
atomic mass
atomic number
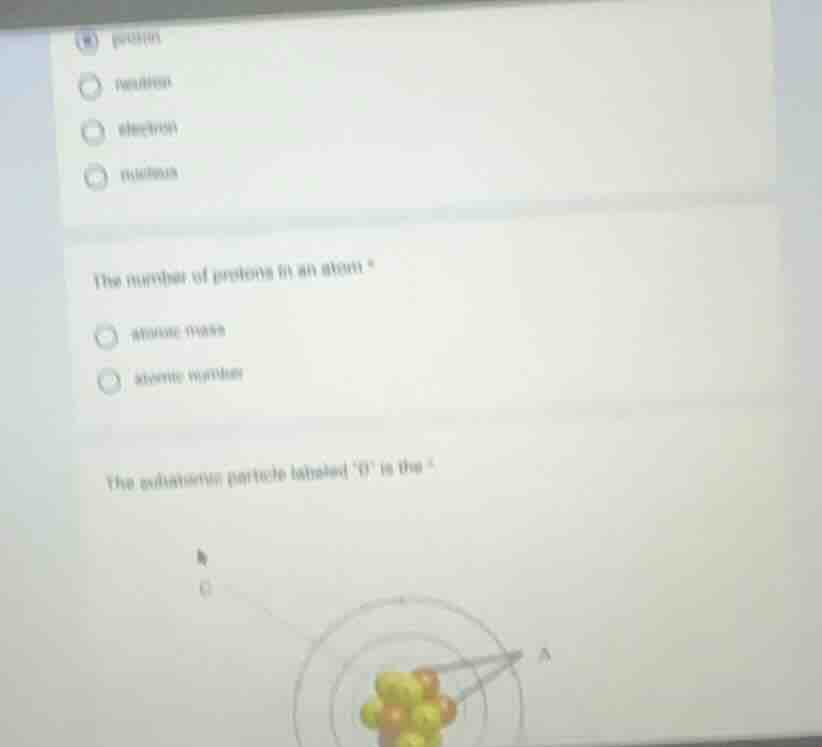
the subatomic particle labeled b is the
First Sub - Question (The number of protons in an atom)
The atomic number of an atom is defined as the number of protons present in the nucleus of that atom. Atomic mass is the sum of the number of protons and neutrons in an atom. So the number of protons in an atom is the atomic number.
In an atomic structure diagram, the particles in the nucleus are protons and neutrons, and the particles outside the nucleus (in electron shells) are electrons. If 'B' is in the nucleus, it could be a proton or a neutron, but if we consider the standard labeling, usually protons (positive charge) and neutrons (neutral) are in the nucleus. If we assume the diagram has protons (positive) and neutrons (neutral) in the nucleus, and given the options (from the first part which had proton, neutron, electron, nucleus), if 'B' is in the nucleus, a common labeled particle in the nucleus is a proton or neutron. But since the first question was about protons and atomic number, and in atomic structure, the nucleus contains protons and neutrons. If we assume the label 'B' is for a proton (since protons are in the nucleus and have a positive charge, and atomic number is based on protons), but without the exact diagram, based on standard atomic structure questions, the subatomic particle in the nucleus (labeled 'B' likely) is a proton or neutron. But from the first part's options (proton, neutron, electron, nucleus), and the nucleus is the region, not a particle. So if 'B' is in the nucleus, the particle is either proton or neutron. But since the first question was about protons, and atomic number is protons, maybe 'B' is a proton. But due to the image being a bit unclear, based on standard atomic structure, the particles in the nucleus are protons (positive) and neutrons (neutral), and electrons are outside. So if 'B' is in the nucleus, the particle is a proton (or neutron, but proton is related to atomic number).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
atomic number (the option corresponding to "atomic number")