QUESTION IMAGE
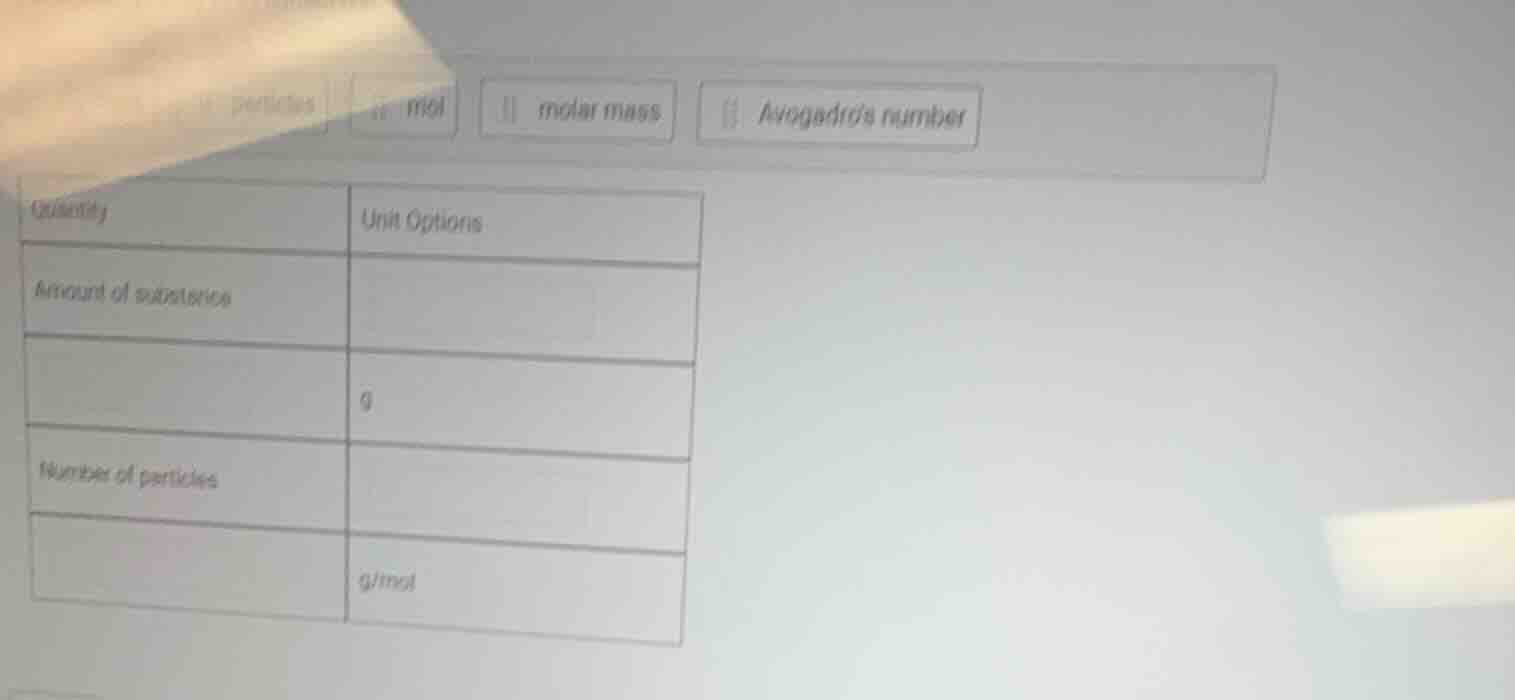
Question
quantity unit options
amount of substance
g
number of particles
g/mol
Step1: Recall unit definitions
- Amount of substance: unit is mol (from the provided terms, "mol" is the unit for amount of substance).
- Mass: unit is g (since "g" is a unit of mass, so the quantity here is mass).
- Number of particles: related to Avogadro's number, and the count of particles can be related to moles via Avogadro's number, but the unit for number of particles is a pure number (or sometimes represented with Avogadro's number context, but from the options, we match the quantity to unit).
- Molar mass: unit is g/mol (molar mass is mass per mole, so unit g/mol).
Step2: Match quantity to unit
- Amount of substance → mol
- Mass → g (so the quantity is mass)
- Number of particles → (related to Avogadro's number, but the unit - less in terms of the given options, but from the terms, "number of particles" is associated with counting, and the unit here would be related to the count, but from the provided boxes, "number of particles" is a quantity, and its unit - related concept is Avogadro's number, but the unit for the amount of substance is mol, mass is g, molar mass is g/mol.
- Molar mass → g/mol
Filling the table:
| Quantity | Unit Options |
|---|---|
| Mass | g |
| Number of particles | (Avogadro's number related, but the quantity is "Number of particles") |
| Molar mass | g/mol |
(If we strictly fill based on the given terms and units):
- Amount of substance: unit is mol (from the "mol" option)
- The quantity with unit g is Mass (since gram is a unit of mass)
- Number of particles: related to the count, and the term "number of particles" is the quantity, with Avogadro's number being the conversion factor, but the unit - for number of particles is a pure number, but from the options, we assign the quantity.
- Molar mass: unit is g/mol (from the "molar mass" term's unit)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Quantity | Unit Options |
|---|---|
| Mass | g |
| Number of particles | (count, related to Avogadro's number) |
| Molar mass | g/mol |
(Or more precisely, filling the blanks in the table as per the columns):
- Amount of substance: mol
- Mass: g (so the quantity is Mass)
- Number of particles: (the quantity is Number of particles, unit - less in terms of the given options but associated with counting)
- Molar mass: g/mol
So the filled table rows (matching quantity to unit):
- Row 1 (Amount of substance): mol
- Row 2 (Mass): g (so quantity is Mass)
- Row 3 (Number of particles): (the quantity is Number of particles)
- Row 4 (Molar mass): g/mol