QUESTION IMAGE
Question
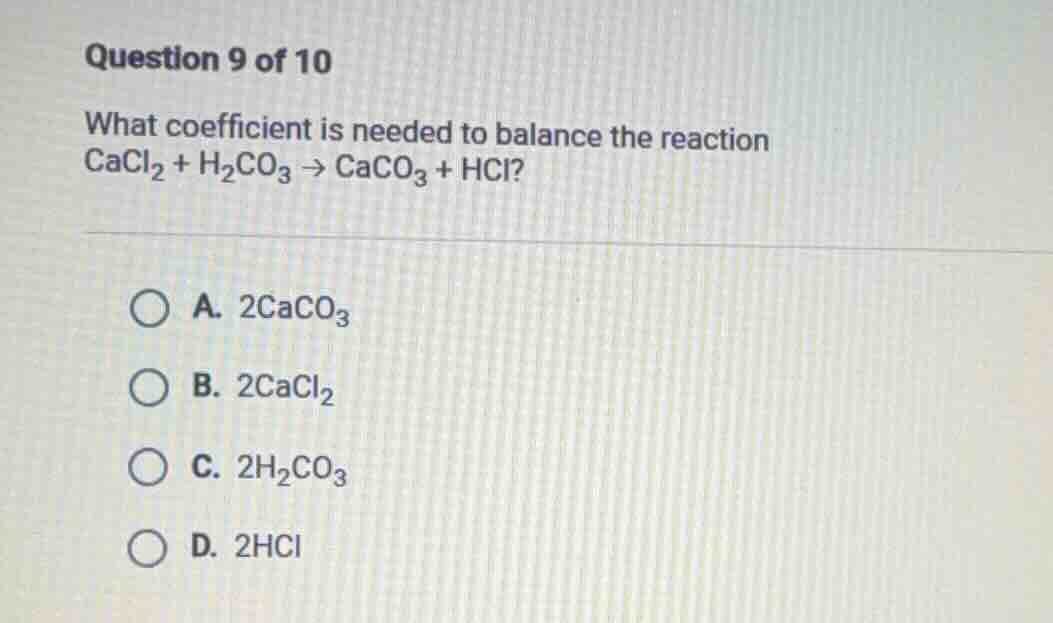
question 9 of 10
what coefficient is needed to balance the reaction
cacl₂ + h₂co₃ → caco₃ + hcl?
a. 2caco₃
b. 2cacl₂
c. 2h₂co₃
d. 2hcl
Step1: Analyze reactants and products
Reactants: \( \text{CaCl}_2 \), \( \text{H}_2\text{CO}_3 \); Products: \( \text{CaCO}_3 \), \( \text{HCl} \)
Step2: Balance each element
- Ca: 1 on left (in \( \text{CaCl}_2 \)), 1 on right (in \( \text{CaCO}_3 \)) – balanced.
- C: 1 on left (in \( \text{H}_2\text{CO}_3 \)), 1 on right (in \( \text{CaCO}_3 \)) – balanced.
- O: 3 on left (in \( \text{H}_2\text{CO}_3 \)), 3 on right (in \( \text{CaCO}_3 \)) – balanced.
- H: 2 on left (in \( \text{H}_2\text{CO}_3 \)), 1 on right (in \( \text{HCl} \)) – need 2 \( \text{HCl} \) to balance H.
- Cl: 2 on left (in \( \text{CaCl}_2 \)), 1 on right (in \( \text{HCl} \)) – 2 \( \text{HCl} \) balances Cl (2 Cl on right).
So adding coefficient 2 to \( \text{HCl} \) balances the equation: \( \text{CaCl}_2 + \text{H}_2\text{CO}_3
ightarrow \text{CaCO}_3 + 2\text{HCl} \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. \( 2\text{HCl} \)