QUESTION IMAGE
Question
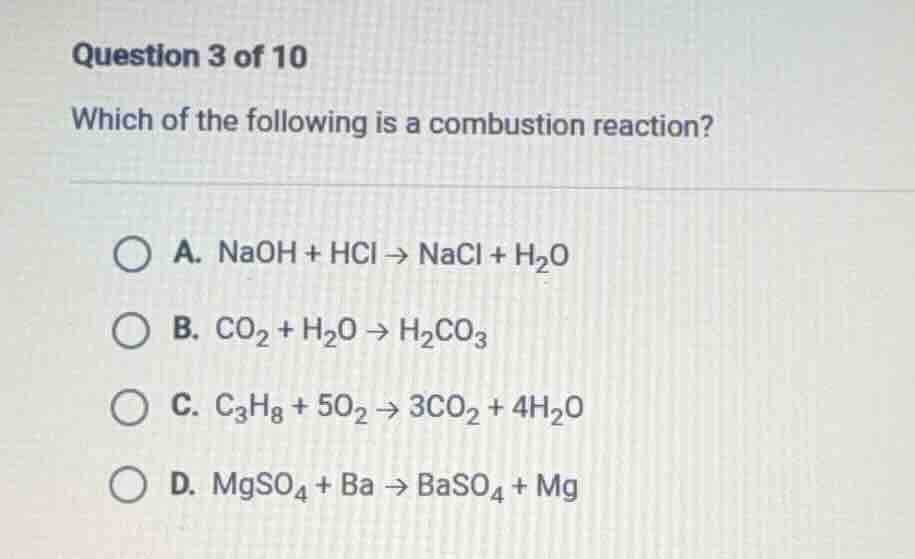
question 3 of 10
which of the following is a combustion reaction?
a. naoh + hcl → nacl + h₂o
b. co₂ + h₂o → h₂co₃
c. c₃h₈ + 5o₂ → 3co₂ + 4h₂o
d. mgso₄ + ba → baso₄ + mg
Brief Explanations
To determine a combustion reaction, we recall that combustion reactions involve a substance reacting with oxygen, producing oxides (usually carbon dioxide and water for hydrocarbon combustion).
- Option A: $\ce{NaOH + HCl -> NaCl + H_{2}O}$ is an acid - base neutralization reaction, not combustion.
- Option B: $\ce{CO_{2} + H_{2}O -> H_{2}CO_{3}}$ is a combination reaction (forming carbonic acid), not combustion.
- Option C: $\ce{C_{3}H_{8} + 5O_{2} -> 3CO_{2} + 4H_{2}O}$ involves propane ($\ce{C_{3}H_{8}}$) reacting with oxygen to produce carbon dioxide and water, which fits the definition of a combustion reaction.
- Option D: $\ce{MgSO_{4} + Ba -> BaSO_{4} + Mg}$ is a single - replacement reaction, not combustion.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. $\ce{C_{3}H_{8} + 5O_{2} -> 3CO_{2} + 4H_{2}O}$