QUESTION IMAGE
Question
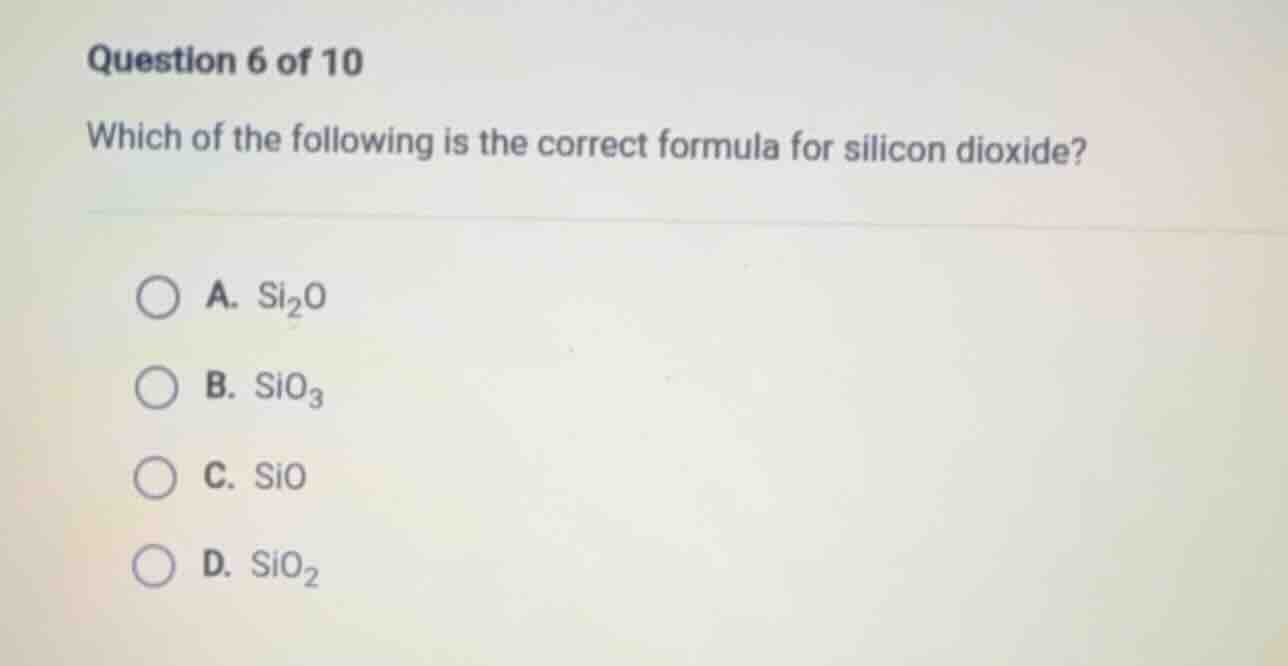
question 6 of 10
which of the following is the correct formula for silicon dioxide?
a. $\ce{si_{2}o}$
b. $\ce{sio_{3}}$
c. $\ce{sio}$
d. $\ce{sio_{2}}$
Brief Explanations
To determine the correct formula for silicon dioxide, we analyze the name. "Silicon" refers to the element Si, and "dioxide" means two (di-) oxygen (oxide) atoms. So the formula should have one Si atom and two O atoms, which is $\ce{SiO_2}$.
- Option A ($\ce{Si_2O}$) has two Si and one O, incorrect.
- Option B ($\ce{SiO_3}$) has one Si and three O, incorrect.
- Option C ($\ce{SiO}$) has one Si and one O, incorrect.
- Option D ($\ce{SiO_2}$) matches the "one Si, two O" from the name "dioxide".
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. $\ce{SiO_2}$