QUESTION IMAGE
Question
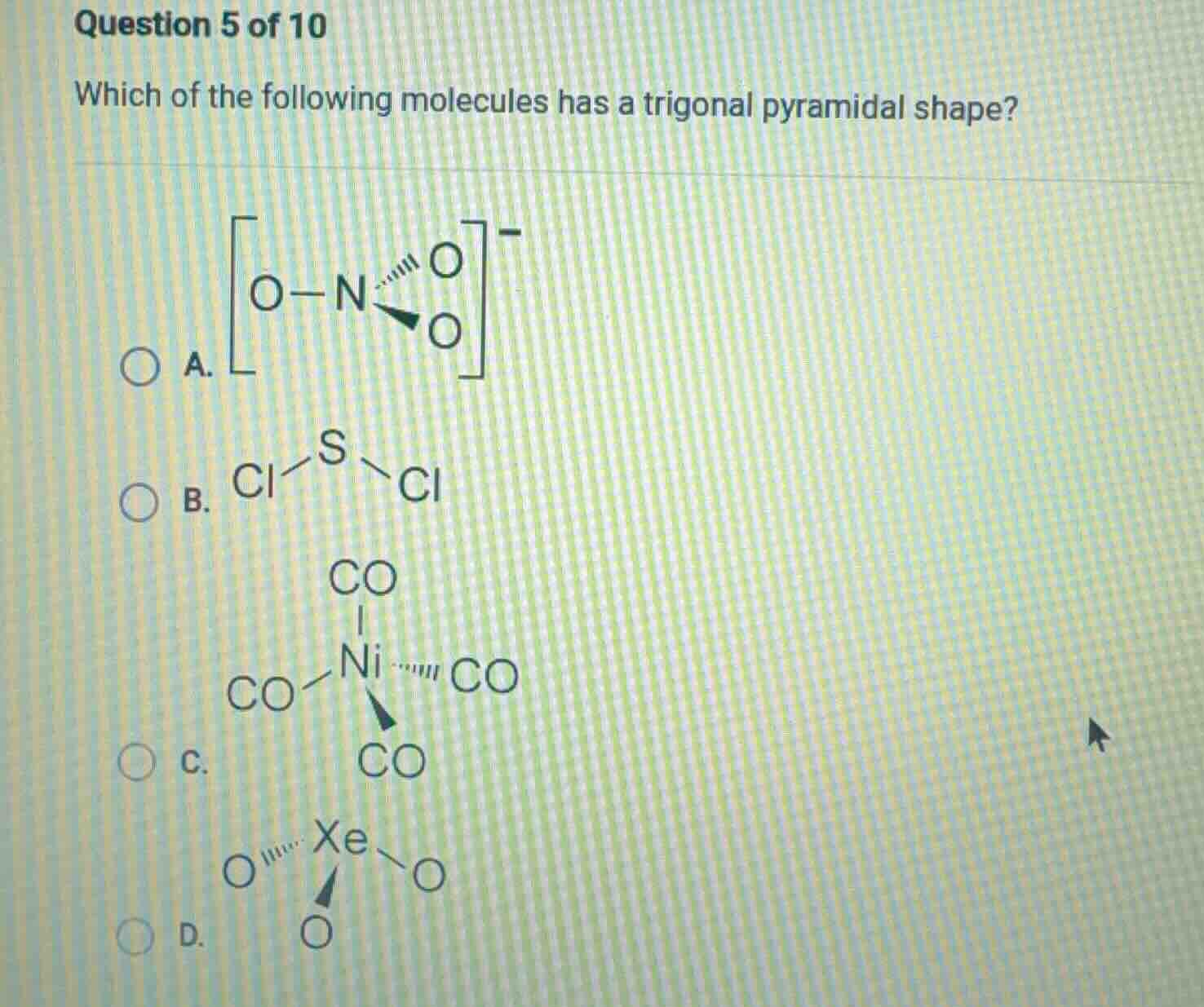
question 5 of 10
which of the following molecules has a trigonal pyramidal shape?
a. \\(\ce{o-n(=o)(-o)^-}\\)
b. \\(\ce{cl-s-cl}\\)
c. \\(\ce{ni(co)_4}\\)
d. \\(\ce{xeo_3}\\)
Brief Explanations
To determine the molecular shape, we analyze the central atom's electron - domain geometry and molecular geometry using VSEPR theory.
- Option A: The central atom is N in $\ce{NO_3^-}$ (the structure shown). The Lewis structure of $\ce{NO_3^-}$ has a central N atom with 3 bonding domains (to O atoms) and no lone pairs (after considering resonance and formal charge). The electron - domain geometry and molecular geometry are trigonal planar.
- Option B: The central atom is S in $\ce{SCl_2}$. The S atom has 2 bonding domains (to Cl atoms) and 2 lone pairs. The electron - domain geometry is tetrahedral, and the molecular geometry is bent (angular).
- Option C: The central atom is Ni in $\ce{Ni(CO)_4}$. The Ni atom has 4 bonding domains (to CO ligands) and no lone pairs. The electron - domain geometry and molecular geometry are tetrahedral.
- Option D: The central atom is Xe in $\ce{XeO_3}$. The Xe atom has 3 bonding domains (to O atoms) and 1 lone pair. According to VSEPR theory, when the central atom has 3 bonding domains and 1 lone pair, the molecular geometry is trigonal pyramidal.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. $\ce{XeO_3}$ (the molecule with central Xe and three O atoms attached)